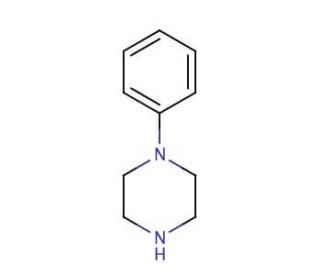
Molecular structure of 1-Phenylpiperazine, CAS Number: 92-54-6
1-Phenylpiperazine (CAS 92-54-6)
Alternate Names:
N-Phenyldiethylenediamine
CAS Number:
92-54-6
Purity:
≥98%
Molecular Weight:
162.23
Molecular Formula:
C10H14N2
Supplemental Information:
This is classified as a Dangerous Good for transport and may be subject to additional shipping charges.
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
1-Phenylpiperazine is a compound that functions as a chemical intermediate in research and development. It acts as a precursor in the synthesis of various organic compounds, including agrochemicals and dyes. Its mode of action involves participating in organic reactions, such as nucleophilic substitution, oxidation, and reduction, to form new chemical entities. Within experimental contexts, 1-Phenylpiperazine serves as a building block for the creation of diverse molecular structures, contributing to the development of novel compounds with applications in various fields.
1-Phenylpiperazine (CAS 92-54-6) References
- SUBSTITUTED 2,3-DIHYDRO-1,5-BENZOTHIAZEPIN-4(5H)-ONES AND 3,4-DIHYDRO-2-PHENYL-(2H)-1,6-BENZOTHIAZOCIN-5(6H)-ONES. | KRAPCHO, J., et al. 1963. J Med Chem. 6: 544-6. PMID: 14173579
- Synthesis, structure and biological activity of 1,2,4-triazolo-1,3-thiazine derivatives. | Foks, H., et al. 1992. Pharmazie. 47: 770-3. PMID: 1480655
- In vitro metabolism studies of (18)F-labeled 1-phenylpiperazine using mouse liver S9 fraction. | Ryu, EK., et al. 2006. Nucl Med Biol. 33: 165-72. PMID: 16546670
- Novel 1-phenylpiperazine and 4-phenylpiperidine derivatives as high-affinity sigma ligands. | Glennon, RA., et al. 1991. J Med Chem. 34: 3360-5. PMID: 1662725
- Novel potent neuropeptide Y Y5 receptor antagonists: synthesis and structure-activity relationships of phenylpiperazine derivatives. | Takahashi, T., et al. 2006. Bioorg Med Chem. 14: 7501-11. PMID: 16919461
- FT-IR and Raman spectroscopic and quantum chemical investigations of some metal halide complexes of 1-phenylpiperazine. | Keşan, G., et al. 2012. Spectrochim Acta A Mol Biomol Spectrosc. 88: 144-55. PMID: 22225603
- DNA-binding, spectroscopic and antimicrobial studies of palladium(II) complexes containing 2,2'-bipyridine and 1-phenylpiperazine. | Shoukry, AA. and Mohamed, MS. 2012. Spectrochim Acta A Mol Biomol Spectrosc. 96: 586-93. PMID: 22864394
- Structure-Function Analysis of Phenylpiperazine Derivatives as Intestinal Permeation Enhancers. | Fein, KC., et al. 2017. Pharm Res. 34: 1320-1329. PMID: 28374339
- ATRP-grown protein-polymer conjugates containing phenylpiperazine selectively enhance transepithelial protein transport. | Cummings, CS., et al. 2017. J Control Release. 255: 270-278. PMID: 28455168
- Alkyl derivatives of tetrahydroisoquinoline, 1-phenylpiperazine, and 4-diphenylmethylpiperidine. | Irwin, WJ., et al. 1972. J Med Chem. 15: 690-2. PMID: 5030942
- [18F]fluoromethylbenzylsulfonate ester: a rapid and efficient synthetic method for the N-[18F]fluoromethylbenzylation of amides and amines. | Choe, YS., et al. 1998. Appl Radiat Isot. 49: 73-7. PMID: 9467837
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
1-Phenylpiperazine, 25 g | sc-224803 | 25 g | $48.00 |
