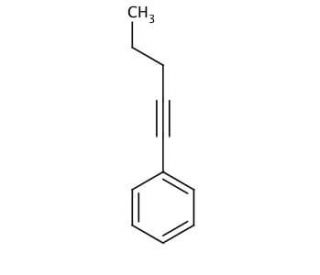
1-Phenyl-1-pentyne (CAS 4250-81-1)
QUICK LINKS
1-Phenyl-1-pentyne is a chemical compound that functions as an alkyne in organic synthesis. It acts as a reactant in various chemical reactions, including Sonogashira coupling, which involves the cross-coupling of aryl halides with terminal alkynes to form substituted alkynes. Participates in the formation of carbon-carbon bonds, playing a role in the construction of complex organic molecules. Its mode of action involves the addition of the alkyne to other organic compounds, leading to the formation of new carbon-carbon bonds. The function in organic synthesis allows for the creation of diverse molecular structures, contributing to the development of new materials and compounds for purposes. Its ability to undergo reactions with different functional groups makes it versatile in the synthesis of organic compounds.
1-Phenyl-1-pentyne (CAS 4250-81-1) References
- An Ammonia Equivalent for the Dimethyltitanocene-Catalyzed Intermolecular Hydroamination of Alkynes. | Haak, E., et al. 2000. Org Lett. 2: 1935-1937. PMID: 10891195
- Ru nanoparticles immobilized on montmorillonite by ionic liquids: a highly efficient heterogeneous catalyst for the hydrogenation of benzene. | Miao, S., et al. 2005. Angew Chem Int Ed Engl. 45: 266-9. PMID: 16311998
- Synthesis of, light emission from, and optical power limiting in soluble single-walled carbon nanotubes functionalized by disubstituted polyacetylenes. | Li, Z., et al. 2006. J Phys Chem B. 110: 2302-9. PMID: 16471817
- Synthesis and light-emitting properties of disubstituted polyacetylenes carrying chromophoric naphthylethynylphenyl pendants. | Lam, JW., et al. 2008. J Phys Chem B. 112: 11227-35. PMID: 18707168
- A highly regioselective synthesis of polysubstituted naphthalene derivatives through gallium trichloride catalyzed alkyne--aldehyde coupling. | Viswanathan, GS., et al. 2002. Angew Chem Int Ed Engl. 41: 2138-41. PMID: 19746621
- Cu-catalyzed arylcarbocyclization of alkynes with diaryliodonium salts through C-C bond formation on inert C(sp3)-H bond. | Peng, J., et al. 2014. Org Lett. 16: 3776-9. PMID: 25008539
- C-H activation/functionalization catalyzed by simple, well-defined low-valent cobalt complexes. | Fallon, BJ., et al. 2015. J Am Chem Soc. 137: 2448-51. PMID: 25625542
- Highly Regio- and Stereoselective Intermolecular Seleno- and Thioamination of Alkynes. | Zheng, G., et al. 2016. Chemistry. 22: 3513-3518. PMID: 26836764
- Controlled Encapsulation of Flower-like Rh-Ni Alloys with MOFs via Tunable Template Dealloying for Enhanced Selective Hydrogenation of Alkyne. | Chen, L., et al. 2016. ACS Appl Mater Interfaces. 8: 31059-31066. PMID: 27783897
- Nickel-Catalyzed Asymmetric [2+2] Cycloaddition Reaction of Hetero-Bicyclic Alkenes with Internal Alkynes. | Qin, H., et al. 2018. Chem Asian J. 13: 2431-2434. PMID: 29968294
- Palladium-Catalyzed Regioselective Heck Coupling of Alkynes with Aryl Iodides for the Synthesis of Trisubstituted Allenes. | Lv, W., et al. 2019. Org Lett. 21: 7795-7798. PMID: 31524415
- Simultaneous determination of Acetaminophen and dopamine based on a water-soluble pillar[6]arene and ultrafine Pd nanoparticle-modified covalent organic framework nanocomposite. | Tan, X., et al. 2021. Analyst. 146: 262-269. PMID: 33118581
- Palladium-catalyzed hydroboration reaction of unactivated alkynes with bis (pinacolato) diboron in water. | Yang, M., et al. 2022. RSC Adv. 12: 9815-9820. PMID: 35424934
- Alkyne insertion into Cu-Al bonds and selective functionalization to form copper acyl compounds. | McManus, C., et al. 2022. Chem Commun (Camb). 58: 8274-8277. PMID: 35790119
- Exploring the Reaction Mechanism of C-H Oxidation by Copper-Salen Complexes. | McKee, ML. 2022. J Phys Chem A. 126: 4969-4980. PMID: 35861503
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
1-Phenyl-1-pentyne, 5 g | sc-224788 | 5 g | $46.00 |
