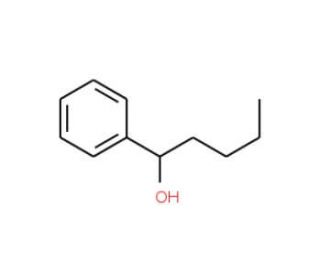
Molecular structure of 1-Phenyl-1-pentanol, CAS Number: 583-03-9
1-Phenyl-1-pentanol (CAS 583-03-9)
Alternate Names:
n-Butyl phenyl carbinol
CAS Number:
583-03-9
Molecular Weight:
164.24
Molecular Formula:
C11H16O
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
1-Phenyl-1-pentanol functions as a chemical intermediate in organic synthesis. It acts as a nucleophile in various reactions, participating in processes such as Grignard reactions and nucleophilic substitution. 1-Phenyl-1-Pentanol can also serve as a chiral auxiliary, facilitating the asymmetric synthesis of organic molecules. At the molecular level, 1-Phenyl-1-pentanol interacts with other reagents to form new carbon-carbon or carbon-heteroatom bonds, contributing to the construction of complex molecular structures. Its mechanism of action involves the formation of intermediates that undergo further transformations to yield desired products.
1-Phenyl-1-pentanol (CAS 583-03-9) References
- Stereoselective 1,2-additions of alpha-alkoxymethyllithiums to aldehydes. | Smyj, RP. and Chong, JM. 2001. Org Lett. 3: 2903-6. PMID: 11529786
- Regioselective Introduction of Electrophiles in the 4-Position of 1-Hydroxypyrazole via Bromine-Lithium Exchange. | Balle, T., et al. 1999. J Org Chem. 64: 5366-5370. PMID: 11674594
- A highly enantioselective phosphabicyclooctane catalyst for the kinetic resolution of benzylic alcohols. | Vedejs, E. and Daugulis, O. 2003. J Am Chem Soc. 125: 4166-73. PMID: 12670239
- Comparison of Chirasil-DEX CB as gas chromatographic and ULMO as liquid chromatographic chiral stationary phase for enantioseparation of aryl- and heteroarylcarbinols. | Uray, G., et al. 2003. J Chromatogr A. 992: 151-7. PMID: 12735471
- Oxidative kinetic resolution of racemic alcohols catalyzed by chiral ferrocenyloxazolinylphosphine-ruthenium complexes. | Nishibayashi, Y., et al. 2003. J Org Chem. 68: 5875-80. PMID: 12868920
- Competing reactions of secondary alcohols with sodium hypochlorite promoted by phase-transfer catalysis. | Bright, ZR., et al. 2005. J Org Chem. 70: 684-7. PMID: 15651819
- Nickel-catalyzed alkylation of aldehydes with trialkylboranes. | Hirano, K., et al. 2005. Org Lett. 7: 4689-91. PMID: 16209511
- Kinetic resolution of racemic secondary alcohols catalyzed by chiral diaminodiphosphine-Ir(I) complexes. | Li, YY., et al. 2006. Org Lett. 8: 5565-7. PMID: 17107073
- Advances in asymmetric oxidative kinetic resolution of racemic secondary alcohols catalyzed by chiral Mn(III) salen complexes. | Ahmad, I., et al. 2017. Chirality. 29: 798-810. PMID: 28963733
- [Experimental study of the choleretic activity of 2'hydroxy-5'carboxy-1-phenyl-1-pentanol]. | Van den Driessche, J., et al. 1968. Therapie. 23: 329-40. PMID: 5701508
- Studies on the interactions of chiral secondary alcohols with rat hydroxysteroid sulfotransferase STa. | Banoglu, E. and Duffel, MW. 1997. Drug Metab Dispos. 25: 1304-10. PMID: 9351908
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
1-Phenyl-1-pentanol, 10 g | sc-273438 | 10 g | $54.00 |
