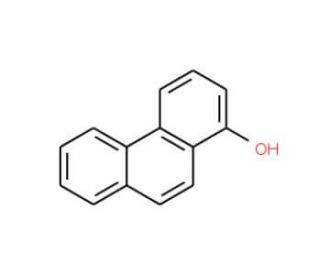
1-Phenanthrol (CAS 2433-56-9)
See product citations (2)
QUICK LINKS
1-Phenanthrol, a polycyclic aromatic compound, has attention in several research domains for its distinct chemical properties and practical applications. In organic chemistry, it serves as a building block for the synthesis of more complex molecules, where its rigid structure is beneficial in the study of molecular conductance and the development of novel organic electronic devices. Environmental scientists employ 1-Phenanthrol in tracing contamination pathways and degradation studies, as it can be a model for understanding the environmental behavior of similar aromatic hydrocarbons. Additionally, it is used in the field of photophysics to explore fluorescence properties, contributing to the design of new luminescent materials. Its interactions with various substrates are also subject to investigation in catalysis research, where it can provide insights into reaction mechanisms and catalyst design.
1-Phenanthrol (CAS 2433-56-9) References
- Protein engineering of cytochrome p450(cam) (CYP101) for the oxidation of polycyclic aromatic hydrocarbons. | Harford-Cross, CF., et al. 2000. Protein Eng. 13: 121-8. PMID: 10708651
- Simultaneous analysis of naphthols, phenanthrols, and 1-hydroxypyrene in urine as biomarkers of polycyclic aromatic hydrocarbon exposure: intraindividual variance in the urinary metabolite excretion profiles caused by intervention with beta-naphthoflavone induction in the rat. | Elovaara, E., et al. 2003. Arch Toxicol. 77: 183-93. PMID: 12698233
- Metabolism of polycyclic compounds. 6. Conversion of phenanthrene into dihydroxydihydrophenanthrenes. | BOYLAND, E. and WOLF, G. 1950. Biochem J. 47: 64-9. PMID: 14791307
- Analysis of phenanthrols in human urine by gas chromatography-mass spectrometry: potential use in carcinogen metabolite phenotyping. | Carmella, SG., et al. 2004. Cancer Epidemiol Biomarkers Prev. 13: 2167-74. PMID: 15598776
- Estrogen stimulates release of secreted amyloid precursor protein from primary rat cortical neurons via protein kinase C pathway. | Zhang, S., et al. 2005. Acta Pharmacol Sin. 26: 171-6. PMID: 15663894
- Metabolism of phenanthrene by the marine cyanobacterium Agmenellum quadruplicatum PR-6. | Narro, ML., et al. 1992. Appl Environ Microbiol. 58: 1351-9. PMID: 1599252
- The effect of the corticosteroid hormone cortexolone on the metabolites produced during phenanthrene biotransformation in Cunninghamella elegans. | Lisowska, K., et al. 2006. Chemosphere. 64: 1499-506. PMID: 16504243
- Production of metabolites in the biodegradation of phenanthrene, fluoranthene and pyrene by the mixed culture of Mycobacterium sp. and Sphingomonas sp. | Zhong, Y., et al. 2011. Bioresour Technol. 102: 2965-72. PMID: 21036605
- Regiospecific oxidation of polycyclic aromatic phenols to quinones by hypervalent iodine reagents. | Wu, A., et al. 2010. Tetrahedron. 66: 2111-2118. PMID: 24014894
- Species-specific metabolism of naphthalene and phenanthrene in 3 species of marine teleosts exposed to Deepwater Horizon crude oil. | Pulster, EL., et al. 2017. Environ Toxicol Chem. 36: 3168-3176. PMID: 28636160
- Evaluation of Interventions to Reduce Firefighter Exposures. | Burgess, JL., et al. 2020. J Occup Environ Med. 62: 279-288. PMID: 31977921
- The effect of alkyl substitution on the oxidative metabolism and mutagenicity of phenanthrene. | Wang, D., et al. 2022. Arch Toxicol. 96: 1109-1131. PMID: 35182162
- Novel metabolites in phenanthrene and pyrene transformation by Aspergillus niger. | Sack, U., et al. 1997. Appl Environ Microbiol. 63: 2906-9. PMID: 9212437
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
1-Phenanthrol, 10 mg | sc-208686 | 10 mg | $380.00 |
