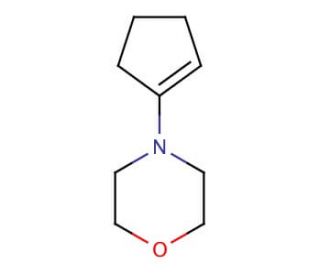
1-Morpholinocyclopentene (CAS 936-52-7)
QUICK LINKS
1-Morpholinocyclopentene is an organic compound. This cyclic amine consists of a five-membered ring and exists as a colorless liquid with a faint odor. 1-Morpholinocyclopentene finds diverse applications, functioning as a solvent, emulsifier, and corrosion inhibitor. In scientific research, it exhibits versatility and finds utility in various applications. It acts as a solvent for organic reactions and serves as a corrosion inhibitor. Additionally, it has been employed in the synthesis of heterocyclic compounds, including oxazolidinones, thiazolidinones, and pyridines. 1-Morpholinocyclopentene, functioning as an organic compound, acts as a base, exhibiting the ability to act as a nucleophile in nucleophilic substitution reactions. Furthermore, it acts as an electron-withdrawing group in electrophilic reactions. Additionally, morpholine can function as a proton acceptor in acid-base reactions.
1-Morpholinocyclopentene (CAS 936-52-7) References
- Asymmetric synthesis of 2-alkyl-perhydroazepines from [5,3,0]-bicyclic lactams. | Meyers, AI., et al. 2001. J Org Chem. 66: 1413-9. PMID: 11312974
- Pi-nucleophilicity in carbon-carbon bond-forming reactions. | Mayr, H., et al. 2003. Acc Chem Res. 36: 66-77. PMID: 12534306
- Antineoplastic Isoflavonoids Derived from Intermediate ortho-Quinone Methides Generated from Mannich Bases. | Frasinyuk, MS., et al. 2016. ChemMedChem. 11: 600-11. PMID: 26889756
- Mesomorphism and Photophysics of Some Metallomesogens Based on Hexasubstituted 2,2':6', 2''-Terpyridines. | Kumar, NS., et al. 2016. Chemistry. 22: 8215-33. PMID: 27138194
- 1,2,4-Triazines in the Synthesis of Bipyridine Bisphenolate ONNO Ligands and Their Highly Luminescent Tetradentate Pt(II) Complexes for Solution-Processable OLEDs. | Pander, P., et al. 2018. Inorg Chem. 57: 3825-3832. PMID: 29537260
- Total Synthesis of (±)-Ginkgolide B. | Corey, EJ., et al. 1988. J Am Chem Soc. 110: 649-651. PMID: 31527923
- 5-Aryl-6-arylthio-2,2'-bipyridine and 6-Arylthio-2,5-diarylpyridine Fluorophores: Pot, Atom, Step Economic (PASE) Synthesis and Photophysical Studies. | Savchuk, MI., et al. 2021. J Fluoresc. 31: 1099-1111. PMID: 33966136
- Contrasting Diastereoselectivity between Cyclic Nitrones and Azomethine Ylides. Stereocontrolled Pathways to cis-anti-anti-cis-Oxazatetraquinanes from a Bicyclic Nitrone. | Reddy, DS., et al. 2021. Org Lett. 23: 5445-5447. PMID: 34210133
- Asymmetrically Functionalized 1,3-Di(2-pyridyl)benzenes: Synthesis and Photophysical Studies. | Starnovskaya, ES., et al. 2022. J Fluoresc. 32: 125-133. PMID: 34618285
- Synthesis and Photophysical Properties of α-(N-Biphenyl)-Substituted 2,2'-Bipyridine-Based Push-Pull Fluorophores. | Starnovskaya, ES., et al. 2022. Molecules. 27: PMID: 36296472
- New TEMPO-Appended 2,2'-Bipyridine-Based Eu(III), Tb(III), Gd(III) and Sm(III) Complexes: Synthesis, Photophysical Studies and Testing Photoluminescence-Based Bioimaging Abilities. | Slovesnova, NV., et al. 2022. Molecules. 27: PMID: 36500504
- Preparation of specifically dideuterated octadecanoates and oxooctadecanoates. | Tulloch, AP. 1977. Lipids. 12: 92-8. PMID: 834126
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
1-Morpholinocyclopentene, 5 g | sc-237586 | 5 g | $77.00 |
