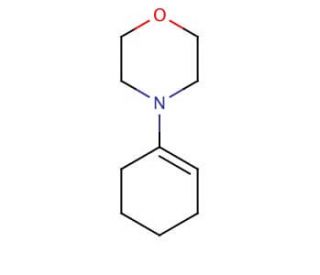
1-Morpholinocyclohexene (CAS 670-80-4)
QUICK LINKS
1-Morpholinocyclohexene is a chemical compound that functions as a versatile building block in organic synthesis. It serves as a key intermediate in the preparation of agrochemicals, and fine chemicals. Its mechanism of action involves participating in a range of chemical reactions, including cycloadditions, nucleophilic additions, and metal-catalyzed transformations. 1-Morpholinocyclohexene′s unique structure and reactivity make it useful for the construction of complex molecular architectures. 1-Morpholinocyclohexene plays a significant role in the synthesis of diverse chemical compounds, contributing to the advancement of various scientific fields. Its ability to undergo selective functionalization and form diverse chemical bonds may be a component in the development of novel molecules with potential applications in a wide range of industries.
1-Morpholinocyclohexene (CAS 670-80-4) References
- Antineoplastic Isoflavonoids Derived from Intermediate ortho-Quinone Methides Generated from Mannich Bases. | Frasinyuk, MS., et al. 2016. ChemMedChem. 11: 600-11. PMID: 26889756
- Phosphonic acid: preparation and applications. | Sevrain, CM., et al. 2017. Beilstein J Org Chem. 13: 2186-2213. PMID: 29114326
- 1-(N-Acylamino)alkyltriarylphosphonium Salts with Weakened Cα-P⁺ Bond Strength-Synthetic Applications. | Adamek, J., et al. 2018. Molecules. 23: PMID: 30257468
- Development of N-F fluorinating agents and their fluorinations: Historical perspective. | Umemoto, T., et al. 2021. Beilstein J Org Chem. 17: 1752-1813. PMID: 34386101
- 1-Aminoalkylphosphonium Derivatives: Smart Synthetic Equivalents of N-Acyliminium-Type Cations, and Maybe Something More: A Review. | Adamek, J., et al. 2022. Molecules. 27: PMID: 35268663
- Michael-Type Addition Reactions of Some Enamines to Arylidenepyrazolones | Abdel-Rahman, M., & Abdel-Ghany, H. 1989. Synthetic Communications. 19(11-12): 1987-1996.
- The electrochemical fluorination of derivatives of morpholine, piperidine and carbazole | Meinert, H., Fackler, R., Mader, J., Reuter, P., & Röhlke, W. 1992. Journal of fluorine chemistry. 59(3): 351-365.
- Reactions of enamines with dehydroaromatic compounds | Gingrich, H. L., Huang, Q., Morales, A. L., & Jones Jr, M. 1992. The Journal of Organic Chemistry. 57(14): 3803-3806.
- Triazolines. Part 32. Synthesis of 1-alkyl-2-aminobenzimidazoles from 5-amino-1-(2-nitroaryl)-1, 2, 3-triazolines. | Erba, E., Mai, G., & Pocar, D. 1992. Journal of the Chemical Society, Perkin Transactions 1. (20): 2709-2712.
- Clay catalyzed synthesis of imines and enamines under solvent-free conditions using microwave irradiation | Varma, R. S., Dahiya, R., & Kumar, S. 1997. Tetrahedron Letters. 38(12): 2039-2042.
- Reaction of α, β-unsaturated trifluoromethyl ketones with cyclic enamines | Nenajdenko, V. G., Druzhinin, S. V., & Balenkova, E. S. 2004. Russian chemical bulletin. 53: 435-442.
- Addition of enamines or pyrroles and B (C6F5) 3 "frustrated Lewis pairs" to alkynes | Dureen, M. A., Brown, C. C., & Stephan, D. W. 2010. Organometallics. 29(23): 6422-6432.
- Reaction of enamines with semicarbazone-based amidoalkylating reagents: A straightforward synthesis of annulated 1-aminopyrimidin-2-one derivatives | Fesenko, A. A., & Shutalev, A. D. 2021. Tetrahedron Letters. 66: 152826.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
1-Morpholinocyclohexene, 25 g | sc-229802 | 25 g | $51.00 |
