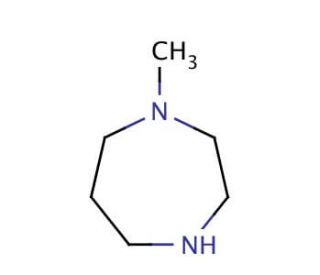
1-Methylhomopiperazine (CAS 4318-37-0)
QUICK LINKS
1-Methylhomopiperazine undergoes coupling reaction with a series of diazonium salts to afford the 4-methyl-1-[2-aryl-1-diazenyl]-1,4-diazepanes. 1-Methylhomopiperazine, a cyclic compound derived from the amino acid homoserine, holds significance in various scientific applications. As a derivative of homopiperazine, a natural cyclic compound with roles in biochemical pathways, it serves as a precursor for essential processes in the human body. 1-Methylhomopiperazine finds utility in the synthesis of diverse compounds, including agrochemicals and polymers. Moreover, it acts as a valuable reagent for the synthesis of heterocyclic compounds like pyridines, pyrimidines, and quinolines. Additionally, this compound demonstrates its versatility as a catalyst for polymer synthesis and as a ligand for the preparation of metal complexes.
1-Methylhomopiperazine (CAS 4318-37-0) References
- Preparation, characterization, and antitumor activity of new cisplatin analogs with homopiperazines: crystal structure of [PtII(1-methylhomopiperazine)(methylmalonato)].2H2O. | Ali, MS., et al. 1999. J Inorg Biochem. 77: 231-8. PMID: 10643661
- Synthesis and biological activities of novel antiallergic agents with 5-lipoxygenase inhibiting action. | Nakano, H., et al. 2000. Bioorg Med Chem. 8: 373-80. PMID: 10722160
- Combinatorial lead optimization of [1,2]-diamines based on ethambutol as potential antituberculosis preclinical candidates. | Lee, RE., et al. 2003. J Comb Chem. 5: 172-87. PMID: 12625709
- Synthesis and structure-activity relationships of 5,6,7,8-tetrahydro-4H-thieno[3,2-b]azepine derivatives: novel arginine vasopressin antagonists. | Cho, H., et al. 2004. J Med Chem. 47: 101-9. PMID: 14695824
- Synthesis of new carbon-11 labeled benzoxazole derivatives for PET imaging of 5-HT(3) receptor. | Gao, M., et al. 2008. Eur J Med Chem. 43: 1570-4. PMID: 18045745
- Protein lysine methyltransferase G9a inhibitors: design, synthesis, and structure activity relationships of 2,4-diamino-7-aminoalkoxy-quinazolines. | Liu, F., et al. 2010. J Med Chem. 53: 5844-57. PMID: 20614940
- Cu(II)-azide polymers with various molar equivalents of blocking diamine ligands: synthesis, structures, magnetic properties with DFT studies. | Mukherjee, S., et al. 2012. Dalton Trans. 41: 54-64. PMID: 21989577
- Design, synthesis and biological evaluation of novel aliphatic amido/sulfonamido-quaternary ammonium salts as antitumor agents. | Song, D., et al. 2013. Bioorg Med Chem. 21: 788-94. PMID: 23266181
- 1-substituted (Dibenzo[b,d]thiophen-4-yl)-2-morpholino-4H-chromen-4-ones endowed with dual DNA-PK/PI3-K inhibitory activity. | Cano, C., et al. 2013. J Med Chem. 56: 6386-401. PMID: 23855836
- Versatility of azide in serendipitous assembly of copper(II) magnetic polyclusters. | Mukherjee, S. and Mukherjee, PS. 2013. Acc Chem Res. 46: 2556-66. PMID: 24246001
- Synthesis and action on the central nervous system of mescaline analogues containing piperazine or homopiperazine rings. | Majchrzak, MW., et al. 1983. J Pharm Sci. 72: 304-6. PMID: 6682439
- Homopiperazine and Its Derivatives. II. A Convenient Synthesis of 1-Methylhomopiperazine | F Popplesdorf, R Myerly, and R Conrow, et al. 1961. J. Org. Chem. 26: 4138–4139.
- Homopiperazine platinum (II) complexes containing substituted disulfide groups: crystal structure of [PtII (homopiperazine)(diphenylsulfide) Cl] NO3 | Mohammad S. Ali a, Uday Mukhopadhyay a, Shervin M. Shirvani a, John Thurston b, Kenton H. Whitmire b, Abdul R. Khokhar a. 2002. Polyhedron. 21: 125-131.
- Synthesis and biological properties of 1,8‐naphthalimidebutylamines. Serotonin 5‐HT1A and 5‐HT7 binding data and pass‐assisted search | Piotr Kowalski, Teresa Kowalska, Andrzej J. Bojarski, Beata Duszyńska. 2007. 44: 889-893.
- Continuous‐flow syntheses of heterocycles | Toma N. Glasnov, C. Oliver Kappe. 2011. 48: 11-30.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
1-Methylhomopiperazine, 5 ml | sc-253938 | 5 ml | $72.00 |
