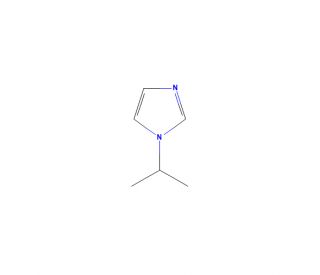
1-Isopropylimidazole (CAS 4532-96-1)
QUICK LINKS
1-Isopropylimidazole (1-IPI) is an organic compound belonging to the imidazole family. This colorless and volatile liquid exhibits a boiling point of 101°C and a melting point of -80°C. With a molecular formula of C4H7N2 and a molecular weight of 87.11 g/mol, 1-Isopropylimidazole plays a significant role in organic synthesis, enabling the preparation of diverse compounds. The scientific research community extensively utilizes 1-Isopropylimidazole due to its wide-ranging applications. Additionally, 1-Isopropylimidazole serves as a valuable reagent in the synthesis of heterocyclic compounds like imidazoles, benzimidazoles, and indoles. Furthermore, its utility extends to the synthesis of polymers, dyes, and catalysts.In chemical reactions, 1-Isopropylimidazole acts as an electrophile, participating in several significant processes. For instance, it partakes in the Knoevenagel condensation, where it reacts with an amine to establish a novel carbon-nitrogen bond. In the Biginelli reaction, it reacts with an aldehyde or ketone to form a fresh carbon-oxygen bond. Finally, in the Fischer indole synthesis, 1-Isopropylimidazole reacts with an amine to generate a novel carbon-nitrogen bond.
1-Isopropylimidazole (CAS 4532-96-1) References
- Abnormal ligand binding and reversible ring hydrogenation in the reaction of imidazolium salts with IrH(5)(PPh(3))(2). | Gründemann, S., et al. 2002. J Am Chem Soc. 124: 10473-81. PMID: 12197749
- A new synthetic route to bulky 'second generation' tris(imidazol-2-ylidene)borate ligands: synthesis of a four coordinate iron(II) complex. | Nieto, I., et al. 2005. Chem Commun (Camb). 3811-3. PMID: 16041425
- An anion-dependent switch in selectivity results from a change of C-H activation mechanism in the reaction of an imidazolium salt with IrH5(PPh3)2. | Appelhans, LN., et al. 2005. J Am Chem Soc. 127: 16299-311. PMID: 16287324
- Experimental and computational investigation of C-N bond activation in ruthenium N-heterocyclic carbene complexes. | Häller, LJ., et al. 2010. J Am Chem Soc. 132: 18408-16. PMID: 21128626
- Synthesis of 1,3-Dialkylimidazol-2-ylidene Boranes from 1,3-Dialkylimidazolium Iodides and Sodium Borohydride. | Gardner, S., et al. 2015. J Org Chem. 80: 9794-7. PMID: 26334670
- Hybrid Gold(I) NHC-Artemether Complexes to Target Falciparum Malaria Parasites. | Ouji, M., et al. 2020. Molecules. 25: PMID: 32570872
- Imidazole-directed fabrication of three polyoxovanadate-based copper frameworks as efficient catalysts for constructing C-N bonds. | Huang, X., et al. 2020. Dalton Trans. 49: 10970-10976. PMID: 32725036
- Inorganic-organic hybrid polyoxovanadates based on [V4O12]4- or [VO3]22- clusters: controllable synthesis, crystal structures and catalytic properties in selective oxidation of sulfides. | Li, J., et al. 2020. Dalton Trans. 49: 14148-14157. PMID: 33021293
- Water-Soluble Pyrene-Adorned Imidazolium Salts with Multicolor Solid-State Fluorescence: Synthesis, Structure, Photophysical Properties, and Application on the Detection of Latent Fingerprints. | Nirmala, M., et al. 2021. ACS Omega. 6: 10318-10332. PMID: 34056185
- Clinically used antifungal azoles as ligands for gold(III) complexes: the influence of the Au(III) ion on the antimicrobial activity of the complex. | Stevanović, NL., et al. 2022. Dalton Trans. 51: 5322-5334. PMID: 35293926
- Vicinal Diaryl-Substituted Isoxazole and Pyrazole Derivatives with In Vitro Growth Inhibitory and In Vivo Antitumor Activity. | Turanlı, S., et al. 2022. ACS Omega. 7: 36206-36226. PMID: 36278052
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
1-Isopropylimidazole, 1 g | sc-460941 | 1 g | $22.00 |
