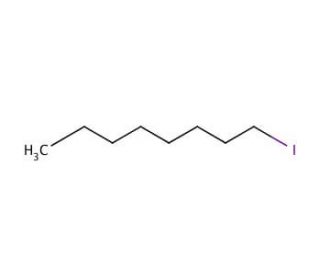
Molecular structure of 1-Iodooctane, CAS Number: 629-27-6
1-Iodooctane (CAS 629-27-6)
CAS Number:
629-27-6
Molecular Weight:
240.13
Molecular Formula:
C8H17I
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
1-Iodooctane is a pyrimidine compound with antimicrobial properties. It reacts with metal surfaces like stainless steel and copper, leading to the production of corresponding hydroxyl groups. Through its interaction with hydrochloric acid, halides are produced, which are potent agents against a spectrum of bacteria, fungi and others. 1-Iodooctane has been used as a reagent to react with polyethylenimine to yield amphiphilic, ultra-sensitive MRI probes. These specialized probes are used in the labeling of chondrocyte cells for further studying.
1-Iodooctane (CAS 629-27-6) References
- Preparation of functionalized aryl iron(II) compounds and a nickel-catalyzed cross-coupling with alkyl halides. | Wunderlich, SH. and Knochel, P. 2009. Angew Chem Int Ed Engl. 48: 9717-20. PMID: 19924751
- The color tuning of PS-b-P2VP lamellar films with changing the alkyl chain length of 1-iodoalkanes. | Shin, SE., et al. 2011. J Nanosci Nanotechnol. 11: 4275-8. PMID: 21780441
- Analytical methodology for determination of organic aerosol functional group distributions. | Kalafut-Pettibone, AJ. and McGivern, WS. 2013. Anal Chem. 85: 3553-60. PMID: 23461576
- Synthesis, Purification and Characterization of Polymerizable Multifunctional Quaternary Ammonium Compounds. | Okeke, UC., et al. 2019. Molecules. 24: PMID: 31013893
- Covalent organic nanospheres as a fiber coating for solid-phase microextraction of genotoxic impurities followed by analysis using GC-MS. | Zhao, Y., et al. 2022. J Pharm Anal. 12: 583-589. PMID: 36105168
- Catalytic reduction of 1,8-diiodooctane by electrogenerated cobalt(I) salen and formation of μ-(1,8-n-octyl)-bis[(salen)cobalt(III)] | Kent S. Alleman, Dennis G. Peters. 1999. Journal of Electroanalytical Chemistry. 460: 207-213.
- Preparation of Polyoxetane Resins Anchoring Pendant Oligo(oxyethylene) Chains and Uses as Polymer Solvents for Alkali Metal Ions | Akihiko Ueyama, Michiko Mizuno, Hiroshi Ogawa, Shigeyoshi Kanoh & Masatoshi Motoi. 2001. Polymer Journal. 33: 754–764.
- Effect of solvent and temperature on the lithium–iodine exchange of primary alkyl iodides: reaction of t-butyllithium with 1-iodooctane in heptane–ether mixtures | William F. Bailey, Jason D. Brubaker, Kevin P. Jordan. 2003. Journal of Organometallic Chemistry. 681: 210-214.
- Alkylation of [2,2′-([2,2′-bipyridine]-6,6′-diyl)bis[phenolato]-N,N′,O,O′]nickel(II) during catalytic reduction of 1-iodooctane | Danielle M Goken, Dennis G Peters, Jonathan A Karty, James P Reilly. 2004. Journal of Electroanalytical Chemistry. 564: 123-132.
- Catalytic reduction of 1-iodooctane by nickel(I) salen electrogenerated at carbon cathodes in dimethylformamide: Effects of added proton donors and a mechanism involving both metal- and ligand-centered one-electron reduction of nickel(II) salen | Philipp W. Raess a, Mohammad S. Mubarak b, Michael A. Ischay a, Matthew P. Foley a, Theodore B. Jennermann a, Krishnan Raghavachari a, Dennis G. Peters a. 2007. Journal of Electroanalytical Chemistry. 603: 124-134.
- Why a cathodic activation by silver interface? Facile reductive homocoupling of 1-iodoalkanes | Philippe Poizot a, Lydia Laffont-Dantras a, Jacques Simonet b. 2008. Electrochemistry Communications. 10: 864-867.
- Synthesis of polymer-supported quaternary ammonium salts and their phase-transfer catalytic activity in halogen-exchange reactions | P. N. Leelamma, K. S. Devaky. 2009. Journal of Applied Polymer Science. 112: 2750-2756.
- Cobalt-catalyzed photopromoted carbonylation of chloroalkanes in the presence of KI | Ying Ping Jia a b, Ying Na Cui b, Jing Mei Yin b, Guang Yun Zhou b, Shen Min Li b, Da Bin Gao b, Xiang Sheng Wang a. 2010. Chinese Chemical Letters. 21: 1033-1036.
- Electrochemistry of substituted salen complexes of nickel(II): Nickel(I)-catalyzed reduction of alkyl and acetylenic halides | Matthew P. Foley a, Peng Du a, Kent J. Griffith a, Jonathan A. Karty a, Mohammad S. Mubarak b, Krishnan Raghavachari a, Dennis G. Peters a. 2010. Journal of Electroanalytical Chemistry. 647: 194-203.
- Synthesis, characterization, and electrochemical study of a new tetradentate nickel(II)-Schiff base complex derived from ethylenediamine and 5′-(N-methyl-N-phenylaminomethyl)-2′-hydroxyacetophenone | Ali Ourari a, Yasmina Ouennoughi a, Djouhra Aggoun a, Mohammad S. Mubarak b, Erick M. Pasciak c, Dennis G. Peters c. 2014. Polyhedron. 67: 59-64.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
1-Iodooctane, 25 g | sc-237573 | 25 g | $62.00 |
