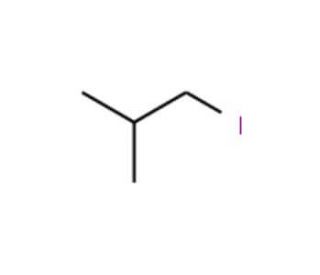
1-Iodo-2-methylpropane (CAS 513-38-2)
QUICK LINKS
1-Iodo-2-methylpropane is in organic synthesis, particularly in the study of alkylation reactions where it acts as an alkylating agent to introduce the isopropyl group into various molecular frameworks. 1-Iodo-2-methylpropane is in the synthesis of more complex organic structures, potentially enhancing the development of new molecules with specific properties. 1-Iodo-2-methylpropane is utilized in reaction mechanisms, helping understand the role of iodine in facilitating nucleophilic substitution reactions. The compound is also explored for its utility in phase transfer catalysis experiments, where it can improve the reaction conditions and yields of organic transformations. 1-Iodo-2-methylpropane plays a role in materials chemistry, particularly in the modification of surface properties of polymers and other materials through grafting techniques.
1-Iodo-2-methylpropane (CAS 513-38-2) References
- Practical asymmetric synthesis of a potent Cathepsin K inhibitor. Efficient palladium removal following Suzuki coupling. | Chen, CY., et al. 2003. J Org Chem. 68: 2633-8. PMID: 12662032
- Asymmetric synthesis of four diastereomers of 3-hydroxy-2,4,6-trimethylheptanoic acid: proof of configurational assignment. | Turk, JA., et al. 2003. J Org Chem. 68: 7841-4. PMID: 14510564
- Rational design of potent and selective NH-linked aryl/heteroaryl cathepsin K inhibitors. | Robichaud, J., et al. 2004. Bioorg Med Chem Lett. 14: 4291-5. PMID: 15261289
- Replacement surgery with unnatural amino acids in the lock-and-key joint of glutathione transferase subunits. | Hegazy, UM., et al. 2006. Chem Biol. 13: 929-36. PMID: 16984882
- Preparation and Characterization of Tetrabenazine Enantiomers against Vesicular Monoamine Transporter 2. | Yu, QS., et al. 2010. ACS Med Chem Lett. 1: 105-109. PMID: 20694189
- Synthesis of cinnamoyl ketoamides as hybrid structures of antioxidants and calpain inhibitors. | Yoo, YJ., et al. 2011. Bioorg Med Chem Lett. 21: 2850-4. PMID: 21504847
- Synchrotron photoionization mass spectrometry measurements of product formation in low-temperature n-butane oxidation: toward a fundamental understanding of autoignition chemistry and n-C4H9 + O2/s-C4H9 + O2 reactions. | Eskola, AJ., et al. 2013. J Phys Chem A. 117: 12216-35. PMID: 24125058
- Using highly emissive and environmentally sensitive o-carborane-functionalized metallophosphors to monitor mitochondrial polarity. | Li, X., et al. 2017. Chem Sci. 8: 5930-5940. PMID: 28989622
- Far-infrared spectra, vibrational assignment, and conformational stability of 1-iodo-2-methylpropane | Durig, J. R., Godbey, S. E., & Sullivan, J. F. 1988. The Journal of Physical Chemistry. 92(24): 6908-6913.
- Microwave, infrared and Raman spectra, conformational stability, barriers to internal rotation, vibrational assignment, and ab initio calculations for 1-fluoro-2-methylpropane | Sullivan, J. F., Wang, A., Durig, J. R., & Godbey, S. E. 1993. Spectrochimica Acta Part A: Molecular Spectroscopy. 49(13-14): 1889-1911.
- Rearrangement and double fluorination in the deiodinative fluorination of neopentyl iodide with xenon difluoride | Patrick, T. B., Zhang, L., & Li, Q. 2000. Journal of Fluorine Chemistry. 102(1-2): 11-15.
- An improved method for cysteine alkylation | Perrey, D. A., & Uckun, F. M. 2001. Tetrahedron Letters. 42(10): 1859-1861.
- Radical yields in the radiolysis of branched hydrocarbons: Tertiary C− H bond rupture in 2, 3-dimethylbutane, 2, 4-dimethylpentane, and 3-ethylpentane | Schuler, R. H., & Wojnarovits, L. 2003. The Journal of Physical Chemistry A. 107(43): 9240-9247.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
1-Iodo-2-methylpropane, 25 g | sc-237572 | 25 g | $85.00 |
