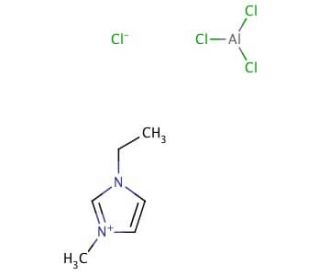
1-Ethyl-3-methylimidazolium tetrachloroaluminate (CAS 80432-05-9)
QUICK LINKS
1-Ethyl-3-methylimidazolium tetrachloroaluminate (EMITCA) represents an intriguing ionic liquid (IL) comprising a 1-ethyl-3-methylimidazolium cation and tetrachloroaluminate anion. This IL holds significant importance due to its exceptional chemical and physical properties, such as its low vapor pressure and remarkable thermal stability. Notably, 1-Ethyl-3-methylimidazolium tetrachloroaluminate finds widespread utility in the realms of both organic and inorganic chemistry, spanning catalysis to analytical applications. Within the domain of organic chemistry, 1-Ethyl-3-methylimidazolium tetrachloroaluminate has emerged as a solvent of choice for synthesizing diverse organic compounds, including β-amino acids, β-amino alcohols, and β-amino esters. Moreover, its role extends as a reaction medium for synthesizing organometallic compounds. In the realm of inorganic chemistry, 1-Ethyl-3-methylimidazolium tetrachloroaluminate serves as a versatile solvent for the synthesis of inorganic compounds such as metal-organic frameworks and metal-organic nanostructures. Additionally, it functions as a reaction medium for the synthesis of various inorganic compounds, including metal oxides and metal sulfides. While the precise mechanism of action for 1-Ethyl-3-methylimidazolium tetrachloroaluminate remains under investigation, it is postulated that the interactions between this IL and the reactants involve hydrogen bonding, electrostatic interactions, and π-π interactions. Additionally, it is believed that 1-Ethyl-3-methylimidazolium tetrachloroaluminate acts as an acid-base catalyst, facilitating the formation of new bonds among the reactants.
1-Ethyl-3-methylimidazolium tetrachloroaluminate (CAS 80432-05-9) References
- Determination of local effects for chloroaluminate ionic liquids on Diels-Alder reactions. | Acevedo, O. 2009. J Mol Graph Model. 28: 95-101. PMID: 19419891
- Capacitively Coupled Plasma Discharge of Ionic Liquid Solutions to Synthesize Carbon Dots as Fluorescent Sensors. | Ke, CB., et al. 2018. Nanomaterials (Basel). 8: PMID: 29861431
- Efficient Approach for the Tritylation of Alcohols Using Recyclable Lewis Acid-Based Ionic Liquid (EMIM·AlCl4). | Chaubey, SA., et al. 2018. ACS Omega. 3: 9607-9612. PMID: 31459091
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
1-Ethyl-3-methylimidazolium tetrachloroaluminate, 100 g | sc-222699 | 100 g | $164.00 | |||
1-Ethyl-3-methylimidazolium tetrachloroaluminate, 1 kg | sc-222699A | 1 kg | $480.00 |
