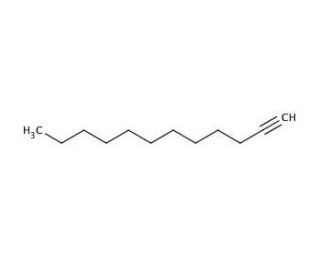
1-Dodecyne (CAS 765-03-7)
QUICK LINKS
1-Dodecyne functions as an alkyne in experimental applications. It acts as a reactive intermediate in various organic synthesis reactions, participating in alkyne metathesis and Sonogashira coupling reactions. Its mechanism of action involves the formation of carbon-carbon bonds through its triple bond, allowing for the construction of complex organic molecules. 1-Dodecyne can serve as a building block for the preparation of functionalized alkyne derivatives, enabling the creation of diverse chemical structures. Its role in experimental applications involves its reactivity as an alkyne, facilitating the synthesis of novel compounds and materials. At the molecular level, 1-Dodecyne interacts with other reagents to form new carbon-carbon bonds, contributing to the development of organic molecules with specific properties.
1-Dodecyne (CAS 765-03-7) References
- Nanopatterning of alkynes on hydrogen-terminated silicon surfaces by scanning probe-induced cathodic electrografting. | Hurley, PT., et al. 2003. J Am Chem Soc. 125: 11334-9. PMID: 16220956
- Intermolecular radical addition of alkylthio- and arylthiodiphenylphosphines to terminal alkynes. | Wada, T., et al. 2008. Org Lett. 10: 1155-7. PMID: 18303899
- Au-catalyzed synthesis of 2-alkylindoles from N-arylhydroxylamines and terminal alkynes. | Wang, Y., et al. 2011. Chem Commun (Camb). 47: 7815-7. PMID: 21637891
- Alkyne-functionalized ruthenium nanoparticles: ruthenium-vinylidene bonds at the metal-ligand interface. | Kang, X., et al. 2012. J Am Chem Soc. 134: 1412-5. PMID: 22229968
- Optimizing P,N-bidentate ligands for oxidative gold catalysis: efficient intermolecular trapping of α-oxo gold carbenes by carboxylic acids. | Ji, K., et al. 2013. Angew Chem Int Ed Engl. 52: 6508-12. PMID: 23640818
- A novel copper containing photoinitiator, copper(II) acylphosphinate, and its application in both the photomediated CuAAC reaction and in atom transfer radical polymerization. | Gong, T., et al. 2013. Chem Commun (Camb). 49: 7950-2. PMID: 23900488
- Single 'click' synthesis of a mixed-mode silica sorbent and application in matrix solid-phase dispersion extraction of β-agonists from porcine liver. | Zhu, Y., et al. 2014. J Chromatogr A. 1354: 101-8. PMID: 24929910
- Rhenium-catalyzed anti-Markovnikov addition reaction of methanetricarboxylates to unactivated terminal acetylenes. | Hori, S., et al. 2015. J Am Chem Soc. 137: 1452-7. PMID: 25563809
- Identification of the formation of metal-vinylidene interfacial bonds of alkyne-capped platinum nanoparticles by isotopic labeling. | Hu, P., et al. 2016. Chem Commun (Camb). 52: 11631-3. PMID: 27538883
- Towards understanding the kinetic behaviour and limitations in photo-induced copper(i) catalyzed azide-alkyne cycloaddition (CuAAC) reactions. | El-Zaatari, BM., et al. 2016. Phys Chem Chem Phys. 18: 25504-25511. PMID: 27711587
- Photopatterned Hydrosilylation on Porous Silicon. | Stewart, MP. and Buriak, JM. 1998. Angew Chem Int Ed Engl. 37: 3257-3260. PMID: 29711412
- The influence of conjugated alkynyl(aryl) surface groups on the optical properties of silicon nanocrystals: photoluminescence through in-gap states. | Angı, A., et al. 2018. Nanotechnology. 29: 355705. PMID: 29862985
- Synthesis of an N-Heterocyclic Carbene-based Au(I) Coordinate Surfactant: Application for Alkyne Hydration Based on Au Nanoparticle Formation. | Taira, T., et al. 2020. J Oleo Sci. 69: 871-882. PMID: 32641614
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
1-Dodecyne, 5 g | sc-224650 | 5 g | $43.00 |
