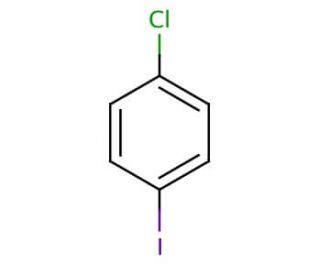
1-Chloro-4-iodobenzene (CAS 637-87-6)
QUICK LINKS
1-Chloro-4-iodobenzene is a chemical compound that functions as a substrate in various organic synthesis reactions. It participates in cross-coupling reactions to form carbon-carbon or carbon-heteroatom bonds, which are in the construction of complex organic molecules. The mode of action of 1-Chloro-4-iodobenzene involves its ability to undergo nucleophilic aromatic substitution reactions, where the chlorine and iodine atoms are replaced by other functional groups. Plays a role in the modification of aromatic compounds, allowing for the introduction of specific substituents at precise positions within the molecular structure. Its reactivity and selectivity make it a useful building block in the synthesis of agrochemicals, and materials with tailored properties. In the development, 1-Chloro-4-iodobenzene serves as a versatile for the creation of diverse organic compounds with potential applications in various fields.
1-Chloro-4-iodobenzene (CAS 637-87-6) References
- Scalable carbon dioxide electroreduction coupled to carbonylation chemistry. | Jensen, MT., et al. 2017. Nat Commun. 8: 489. PMID: 28887452
- Transition-Metal-Free Radical C(sp3)-C(sp2) and C(sp3)-C(sp3) Coupling Enabled by 2-Azaallyls as Super-Electron-Donors and Coupling-Partners. | Li, M., et al. 2017. J Am Chem Soc. 139: 16327-16333. PMID: 29019654
- Synthesis of 3-Alkylideneisoindolin-1-ones via Sonogashira Cyclocarbonylative Reactions of 2-Ethynylbenzamides. | Albano, G., et al. 2020. J Org Chem. 85: 10022-10034. PMID: 32615762
- Transition metal-catalysed A-ring C-H activations and C(sp2)-C(sp2) couplings in the 13α-oestrone series and in vitro evaluation of antiproliferative properties. | Traj, P., et al. 2021. J Enzyme Inhib Med Chem. 36: 895-902. PMID: 33771084
- Ni(II) Precatalysts Enable Thioetherification of (Hetero)Aryl Halides and Tosylates and Tandem C-S/C-N Couplings. | Martín, MT., et al. 2021. Chemistry. 27: 12320-12326. PMID: 34191385
- Photoredox-Catalyzed Reduction of Halogenated Arenes in Water by Amphiphilic Polymeric Nanoparticles. | Eisenreich, F., et al. 2021. Molecules. 26: PMID: 34641426
- Selective hydroxylation of aryl iodides to produce phenols under mild conditions using a supported copper catalyst. | Hao, L., et al. 2021. RSC Adv. 11: 25348-25353. PMID: 35478897
- Copper-catalysed enantioselective intramolecular etherification of propargylic esters: synthetic approach to chiral isochromans. | Liu, S., et al. 2019. RSC Adv. 9: 18918-18922. PMID: 35516894
- Palladium nanoparticles on a pyridinium supported ionic liquid phase: a recyclable and low-leaching palladium catalyst for aminocarbonylation reactions. | Adamcsik, B., et al. 2020. RSC Adv. 10: 23988-23998. PMID: 35517315
- Phenylboronic Ester-Activated Aryl Iodide-Selective Buchwald-Hartwig-Type Amination toward Bioactivity Assay. | Dhital, RN., et al. 2022. ACS Omega. 7: 24184-24189. PMID: 35874269
- A One-Pot Synthesis-Functionalization Strategy for Streamlined Access to 2,5-Disubstituted 1,3,4-Oxadiazoles from Carboxylic Acids. | Matheau-Raven, D. and Dixon, DJ. 2022. J Org Chem. 87: 12498-12505. PMID: 36054913
- Intraligand Charge Transfer Enables Visible-Light-Mediated Nickel-Catalyzed Cross-Coupling Reactions. | Cavedon, C., et al. 2022. Angew Chem Int Ed Engl. 61: e202211433. PMID: 36161982
- Dimsyl Anion Enables Visible-Light-Promoted Charge Transfer in Cross-Coupling Reactions of Aryl Halides. | Pan, L., et al. 2022. Adv Synth Catal. 364: 420-425. PMID: 37197314
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
1-Chloro-4-iodobenzene, 25 g | sc-224615 | 25 g | $37.00 |
