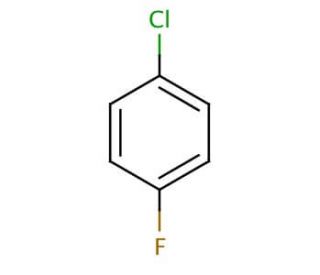
1-Chloro-4-fluorobenzene (CAS 352-33-0)
QUICK LINKS
1-Chloro-4-fluorobenzene (1-Cl-4-F-benzene) is an aromatic chemical compound that possesses distinct characteristics. It presents itself as a colorless liquid with a pungent odor and exhibits high flammability. Its wide array of applications extends to both scientific research and industrial sectors, where it serves as a precursor to other organic compounds and a reagent in chemical reactions. 1-Chloro-4-fluorobenzene plays a vital role in numerous scientific research applications. It acts as a reagent in chemical reactions, particularly in the synthesis of compound derivatives. Furthermore, this compound serves as a key building block for synthesizing diverse organic compounds, including heterocyclic compounds. The mechanism of action of 1-Chloro-4-fluorobenzene involves its role as a Lewis acid during chemical reactions. It forms an intermediate complex with the halide, abstracting the halide from the reaction mixture.
1-Chloro-4-fluorobenzene (CAS 352-33-0) References
- Multiresidue analysis of pesticides in fruits and vegetables using solid-phase extraction and gas chromatographic methods. | Lal, A., et al. 2008. Anal Sci. 24: 231-6. PMID: 18270414
- Ni(I) and Ni(II) ring-expanded N-heterocyclic carbene complexes: C-H activation, indole elimination and catalytic hydrodehalogenation. | Davies, CJ., et al. 2010. Chem Commun (Camb). 46: 5151-3. PMID: 20559596
- Complete hydrodehalogenation of polyfluorinated and other polyhalogenated benzenes under mild catalytic conditions. | Baumgartner, R., et al. 2013. Environ Sci Technol. 47: 6545-53. PMID: 23663092
- Palladium-catalyzed direct intermolecular α-arylation of amides with aryl chlorides. | Zheng, B., et al. 2013. Org Lett. 15: 4190-3. PMID: 23909856
- BippyPhos: a single ligand with unprecedented scope in the Buchwald-Hartwig amination of (hetero)aryl chlorides. | Crawford, SM., et al. 2013. Chemistry. 19: 16760-71. PMID: 24281816
- Borylation of unactivated aryl chlorides under mild conditions by using diisopropylaminoborane as a borylating reagent. | Guerrand, HD., et al. 2014. Chemistry. 20: 5573-9. PMID: 24729439
- A General and Practical Palladium-Catalyzed Direct α-Arylation of Amides with Aryl Halides. | Zheng, B., et al. 2014. Adv Synth Catal. 356: 165-178. PMID: 24765060
- Regioselective synthesis of benzimidazolones via cascade C-N coupling of monosubstituted ureas. | Ernst, JB., et al. 2014. Org Lett. 16: 3844-6. PMID: 24971635
- High-performance heterogeneous catalysis with surface-exposed stable metal nanoparticles. | Huang, N., et al. 2014. Sci Rep. 4: 7228. PMID: 25427425
- Palladium-catalyzed α-arylation of aryl acetic acid derivatives via dienolate intermediates with aryl chlorides and bromides. | Sha, SC., et al. 2015. Org Lett. 17: 410-3. PMID: 25582024
- Palladium-catalyzed C(sp(3))-H Arylation of N-Boc benzylalkylamines via a deprotonative cross-coupling process. | Hussain, N., et al. 2015. Chemistry. 21: 11010-3. PMID: 26129922
- Palladium-Catalyzed Direct C3-Selective Arylation of N-Unsubstituted Indoles with Aryl Chlorides and Triflates. | Yamaguchi, M., et al. 2017. Org Lett. 19: 5388-5391. PMID: 28898099
- Electrophotocatalytic SN Ar Reactions of Unactivated Aryl Fluorides at Ambient Temperature and Without Base. | Huang, H. and Lambert, TH. 2020. Angew Chem Int Ed Engl. 59: 658-662. PMID: 31583795
- Nickel-catalyzed cross-electrophile coupling of aryl chlorides with allylic alcohols. | Yu, H. and Wang, ZX. 2021. Org Biomol Chem. 19: 9723-9731. PMID: 34727149
- Photoinduced FeCl3-Catalyzed Chlorination of Aromatic Sulfonyl Chloride via Extrusion of SO2 at Room Temperature. | Deng, M., et al. 2023. Org Lett. 25: 4576-4580. PMID: 37289092
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
1-Chloro-4-fluorobenzene, 25 g | sc-237522 | 25 g | $47.00 |
