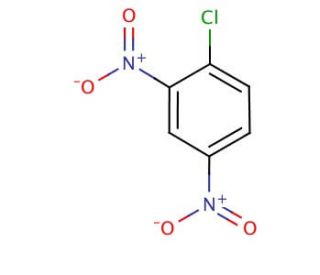
Molecular structure of 1-Chloro-2,4-dinitrobenzene, CAS Number: 97-00-7
1-Chloro-2,4-dinitrobenzene (CAS 97-00-7)
See product citations (1)
Alternate Names:
Dinitrochlorobenzene; 2,4-Dinitrochlorobenzene; DNC; CDNB
Application:
1-Chloro-2,4-dinitrobenzene is a reagent for the detection and determination of pyridine compounds
CAS Number:
97-00-7
Purity:
≥98%
Molecular Weight:
202.55
Molecular Formula:
C6H3ClN2O4
Supplemental Information:
This is as a Dangerous Good for transport and may be subject to additional shipping charges.
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Product Citations
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
1-Chloro-2,4-dinitrobenzene is a substrate of GST. It has been used as alkylating agent to evaluate the depletion of intracellular erythrocyte glutathione (GSH). 1-Chloro-2,4-dinitrobenzene on incubation with erythrocyte undergoes conjugation with erythrocyte glutathione (GSH) to form 2,4-dinitrophenyl-S-glutathione. It is an irreversible inhibitor of human thioredoxin reductase. Additionally, it is a skin irritant that may cause dermatitis of both primary and allergic types. Contact sensitization with the product has been used as a measure of cellular immunity. Also, 1-chloro-2,4-dinitrobenzene is used as a reagent for the detection and determination of pyridine compounds.
1-Chloro-2,4-dinitrobenzene (CAS 97-00-7) References
- Detoxification of 1-chloro-2,4-dinitrobenzene in MCF7 breast cancer cells expressing glutathione S-transferase P1-1 and/or multidrug resistance protein 1. | Diah, SK., et al. 1999. Toxicol Appl Pharmacol. 157: 85-93. PMID: 10366541
- Expression of hGSTP1 alleles in human lung and catalytic activity of the native protein variants towards 1-chloro-2,4-dinitrobenzene, 4-vinylpyridine and (+)-anti benzo[a]pyrene-7,8-diol-9,10-oxide. | Coles, B., et al. 2000. Cancer Lett. 156: 167-75. PMID: 10880766
- Effect of 1-chloro-2,4-dinitrobenzene on K+ transport in normal and sickle human red blood cells. | Muzyamba, MC. and Gibson, JS. 2003. J Physiol. 547: 903-11. PMID: 12576491
- Growth and characterization of organic nonlinear optical crystal of 1-chloro-2,4-dinitrobenzene (CDNB). | Sethuraman, K., et al. 2007. Spectrochim Acta A Mol Biomol Spectrosc. 66: 707-11. PMID: 16875866
- Molecular determinants of xenobiotic metabolism: QM/MM simulation of the conversion of 1-chloro-2,4-dinitrobenzene catalyzed by M1-1 glutathione S-transferase. | Bowman, AL., et al. 2007. Biochemistry. 46: 6353-63. PMID: 17480056
- Analysis of vibrational spectra of 1-chloro-2,4-dinitrobenzene based on density functional theory calculations. | Krishnakumar, V. and Prabavathi, N. 2009. Spectrochim Acta A Mol Biomol Spectrosc. 72: 738-42. PMID: 19124269
- Intrahepatic conversion of a glutathione conjugate to its mercapturic acid. Metabolism of 1-chloro-2,4-dinitrobenzene in isolated perfused rat and guinea pig livers. | Hinchman, CA., et al. 1991. J Biol Chem. 266: 22179-85. PMID: 1939239
- Effect of dehydroepiandrosterone on atopic dermatitis-like skin lesions induced by 1-chloro-2,4-dinitrobenzene in mouse. | Chan, CC., et al. 2013. J Dermatol Sci. 72: 149-57. PMID: 23891346
- Synthesis of a new magnetic-MIP for the selective detection of 1-chloro-2,4-dinitrobenzene, a highly allergenic compound. | Uzuriaga-Sánchez, RJ., et al. 2017. Mater Sci Eng C Mater Biol Appl. 74: 365-373. PMID: 28254306
- Glutathione S-conjugate formation from 1-chloro-2,4-dinitrobenzene and biliary S-conjugate excretion in the perfused rat liver. | Wahlländer, A. and Sies, H. 1979. Eur J Biochem. 96: 441-6. PMID: 467417
- Cross specificity in some vertebrate and insect glutathione-transferases with methyl parathion (dimethyl p-nitrophenyl phosphorothionate), 1-chloro-2,4-dinitro-benzene and s-crotonyl-N-acetylcysteamine as substrates. | Clark, AG., et al. 1973. Biochem J. 135: 385-92. PMID: 4772267
- Enzymatic conjugation of erythrocyte glutathione with 1-chloro-2,4-dinitrobenzene: the fate of glutathione conjugate in erythrocytes and the effect of glutathione depletion on hemoglobin. | Awasthi, YC., et al. 1981. Blood. 58: 733-8. PMID: 7272504
- 1-Chloro-2,4-dinitrobenzene is an irreversible inhibitor of human thioredoxin reductase. Loss of thioredoxin disulfide reductase activity is accompanied by a large increase in NADPH oxidase activity. | Arnér, ES., et al. 1995. J Biol Chem. 270: 3479-82. PMID: 7876079
- Measurement and characterization of the denitrosation of tauromustine and related nitrosoureas by glutathione transferases in liver cytosol from various species. | Tuvesson, H., et al. 1993. Carcinogenesis. 14: 1143-7. PMID: 8508500
- Glutathione conjugation with 1-chloro-2,4-dinitrobenzene (CDNB): interindividual variability in human liver, lung, kidney and intestine. | Temellini, A., et al. 1995. Int J Clin Pharmacol Ther. 33: 498-503. PMID: 8520807
Substrate of:
GST.Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
1-Chloro-2,4-dinitrobenzene, 1 g | sc-237519 | 1 g | $36.00 | |||
1-Chloro-2,4-dinitrobenzene, 10 g | sc-237519B | 10 g | $51.00 | |||
1-Chloro-2,4-dinitrobenzene, 100 g | sc-237519A | 100 g | $70.00 | |||
1-Chloro-2,4-dinitrobenzene, 500 g | sc-237519C | 500 g | $315.00 |
