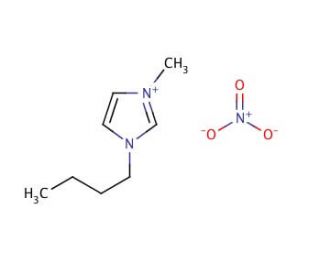
1-Butyl-3-methylimidazolium nitrate (CAS 179075-88-8)
QUICK LINKS
1-Butyl-3-methylimidazolium nitrate has found extensive application as a solvent in various reactions, including condensation, nucleophilic substitution, and oxidation reactions. Within catalysis, it has proven effective in facilitating ester hydrolysis and polymer synthesis. In the realm of electrochemical processes, 1-Butyl-3-methylimidazolium nitrate serves as an electrolyte for the electrochemical oxidation of organic compounds. The distinctive properties of this compound can be attributed to the presence of the imidazolium cation and the nitrate anion. The imidazolium cation is recognized for its Lewis base properties, while the nitrate anion exhibits Lewis acid characteristics. This combination of Lewis acids and bases contributes to the exceptional thermal and chemical stability of 1-Butyl-3-methylimidazolium nitrate, as well as its low volatility.
1-Butyl-3-methylimidazolium nitrate (CAS 179075-88-8) References
- Precipitation of a dioxouranium(VI) species from a room temperature ionic liquid medium. | Bradley, AE., et al. 2002. Inorg Chem. 41: 1692-4. PMID: 11925156
- Parametrization of 1-butyl-3-methylimidazolium hexafluorophosphate/nitrate ionic liquid for the GROMOS force field. | Micaelo, NM., et al. 2006. J Phys Chem B. 110: 14444-51. PMID: 16854154
- Protein structure and dynamics in ionic liquids. Insights from molecular dynamics simulation studies. | Micaêlo, NM. and Soares, CM. 2008. J Phys Chem B. 112: 2566-72. PMID: 18266354
- Confinement of 1-butyl-3-methylimidazolium nitrate in metallic silver. | Neouze, MA. and Litschauer, M. 2008. J Phys Chem B. 112: 16721-5. PMID: 19367985
- Investigation of ionic liquids under Bi-ion and Bi-cluster ions bombardment by ToF-SIMS. | Holzweber, M., et al. 2010. J Mass Spectrom. 45: 1104-10. PMID: 20690159
- [Adsorption performances for sulfur dioxide using imidazole ionic liquids]. | Tang, YX., et al. 2010. Huan Jing Ke Xue. 31: 2582-6. PMID: 21250436
- New insight into molecular interactions of imidazolium ionic liquids with bovine serum albumin. | Shu, Y., et al. 2011. J Phys Chem B. 115: 12306-14. PMID: 21919506
- Ionic liquid-induced formation of the α-helical structure of β-lactoglobulin. | Takekiyo, T., et al. 2013. J Phys Chem B. 117: 10142-8. PMID: 23926920
- Anomalous Freezing of Nano-Confined Water in Room-Temperature Ionic Liquid 1-Butyl-3-Methylimidazolium Nitrate. | Abe, H., et al. 2016. Chemphyschem. 17: 1136-42. PMID: 26841088
- Methanol-Promoted Oxidation of Nitrogen Oxide (NOx) by Encapsulated Ionic Liquids. | Santiago, R., et al. 2019. Environ Sci Technol. 53: 11969-11978. PMID: 31518126
- Molecular Dynamics Insights into the Nanoscale Structural Organization and Local Interaction of Aqueous Solutions of Ionic Liquid 1-Butyl-3-methylimidazolium Nitrate. | Kowsari, MH. and Torabi, SM. 2020. J Phys Chem B. 124: 6972-6985. PMID: 32687363
- Effect of Imidazolium Nitrate Ionic Liquids on Conformational Changes of Poly(N-vinylcaprolactam). | Umapathi, R., et al. 2022. ACS Omega. 7: 39742-39749. PMID: 36385857
- Effects of 1-butyl-3-methylimidazolium nitrate on the thermal hazardous properties of lignitous and long flame coal through a green approach and thermokinetic models | Laiwang, B., Tsai, Y. T., Liu, S. H., Deng, J., Xiao, Y., Wang, Q. H., & Shu, C. M. (2019). Process Safety and Environmental Protection,. 131,: 127-134.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
1-Butyl-3-methylimidazolium nitrate, 5 g | sc-222683 | 5 g | $238.00 | |||
1-Butyl-3-methylimidazolium nitrate, 50 g | sc-222683A | 50 g | $702.00 |
