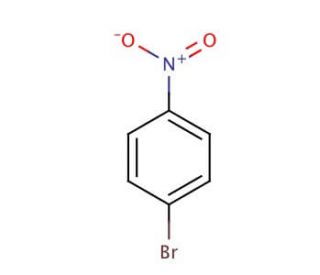
1-Bromo-4-nitrobenzene (CAS 586-78-7)
QUICK LINKS
1-Bromo-4-nitrobenzene is a chemical compound that functions as an electrophilic aromatic substitution reagent in organic synthesis. It acts as a source of the nitro group in various reactions, allowing for the introduction of this functional group into aromatic compounds. The mechanism of action of 1-Bromo-4-nitrobenzene involves the electrophilic substitution of the bromine atom for a hydrogen atom on the aromatic ring, followed by the subsequent substitution of the nitro group for the bromine atom. This process results in the formation of a nitrobenzene derivative, which can then be further modified or utilized in the synthesis of more complex organic molecules. In this way, 1-Bromo-4-nitrobenzene plays a specific role in the construction of diverse chemical structures, contributing to the development of new compounds for various development purposes.
1-Bromo-4-nitrobenzene (CAS 586-78-7) References
- Polyurea-encapsulated palladium(II) acetate: a robust and recyclable catalyst for use in conventional and supercritical media. | Ley, SV., et al. 2002. Chem Commun (Camb). 1134-5. PMID: 12122701
- Efficient Stille cross-coupling reaction catalyzed by the Pd(OAc)2/Dabco catalytic system. | Li, JH., et al. 2005. J Org Chem. 70: 2832-4. PMID: 15787581
- Reusable copper-catalyzed cross-coupling reactions of aryl halides with organotins in inexpensive ionic liquids. | Li, JH., et al. 2006. J Org Chem. 71: 7488-90. PMID: 16958550
- Pd(OAc)2-catalyzed fluoride-free cross-coupling reactions of arylsiloxanes with aryl bromides in aqueous medium. | Shi, S. and Zhang, Y. 2007. J Org Chem. 72: 5927-30. PMID: 17585827
- Direct palladium-catalyzed arylations of aryl bromides with 2/9-substituted pyrimido[5,4-b]indolizines. | Jiang, M., et al. 2009. J Comb Chem. 11: 806-8. PMID: 19645499
- Gelatin as a bioorganic reductant, ligand and support for palladium nanoparticles. Application as a catalyst for ligand- and amine-free Sonogashira-Hagihara reaction. | Firouzabadi, H., et al. 2011. Org Biomol Chem. 9: 865-71. PMID: 21120230
- A highly active and magnetically retrievable nanoferrite-DOPA-copper catalyst for the coupling of thiophenols with aryl halides. | Baig, RB. and Varma, RS. 2012. Chem Commun (Camb). 48: 2582-4. PMID: 22293995
- Changed reactivity of the 1-bromo-4-nitrobenzene radical anion in a room temperature ionic liquid. | Ernst, S., et al. 2013. Phys Chem Chem Phys. 15: 6382-9. PMID: 23525257
- The electrochemical reduction of 1-bromo-4-nitrobenzene at zinc electrodes in a room-temperature ionic liquid: a facile route for the formation of arylzinc compounds. | Ernst, S., et al. 2014. Phys Chem Chem Phys. 16: 4478-82. PMID: 24473222
- Highly ordered surfactant micelles function as the extraction matrix for direct electrochemical detection of halonitrobenzenes at the ppb level. | Yao, L., et al. 2016. Analyst. 141: 2303-7. PMID: 26887641
- Biological evaluation and molecular docking studies of nitro benzamide derivatives with respect to in vitro anti-inflammatory activity. | Tumer, TB., et al. 2017. Int Immunopharmacol. 43: 129-139. PMID: 27988460
- Phosphine-Based Covalent Organic Framework for the Controlled Synthesis of Broad-Scope Ultrafine Nanoparticles. | Tao, R., et al. 2020. Small. 16: e1906005. PMID: 31971660
- New Amphiphilic Imidazolium/Benzimidazolium Calix[4]arene Derivatives: Synthesis, Aggregation Behavior and Decoration of DPPC Vesicles for Suzuki Coupling in Aqueous Media. | Burilov, V., et al. 2020. Nanomaterials (Basel). 10: PMID: 32532131
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
1-Bromo-4-nitrobenzene, 5 g | sc-237492 | 5 g | $22.00 | |||
1-Bromo-4-nitrobenzene, 25 g | sc-237492A | 25 g | $56.00 |
