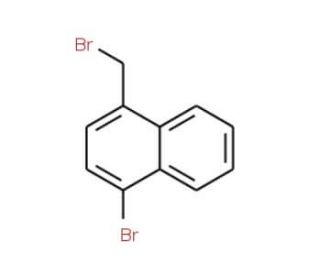
1-Bromo-4-(bromomethyl)naphthalene (CAS 79996-99-9)
QUICK LINKS
1-Bromo-4-(bromomethyl)naphthalene is a chemical compound that has attention in the field of organic chemistry, particularly for its role as an intermediate in the synthesis of various polycyclic aromatic hydrocarbons. Its bifunctional nature, with two reactive bromine sites, makes it a valuable building block in the construction of more complex molecules through cross-coupling reactions, a topic of intensive study for creating new materials and catalysts. Additionally, researchers utilize this compound to investigate the mechanisms of electrophilic aromatic substitution reactions, which are fundamental to the synthesis of many organic compounds. The study of such halogenated naphthalenes also contributes to the understanding of the environmental impact and behavior of similar compounds.
1-Bromo-4-(bromomethyl)naphthalene (CAS 79996-99-9) References
- High physiological thermal triplex stability optimization of twisted intercalating nucleic acids (TINA). | Bomholt, N., et al. 2008. Org Biomol Chem. 6: 3714-22. PMID: 18843401
- Effects of electronics, aromaticity, and solvent polarity on the rate of azaquinone-methide-mediated depolymerization of aromatic carbamate oligomers. | Robbins, JS., et al. 2013. J Org Chem. 78: 3159-69. PMID: 23414397
- Discovery of Novel Bicyclic Imidazolopyridine-Containing Human Urate Transporter 1 Inhibitors as Hypouricemic Drug Candidates with Improved Efficacy and Favorable Druggability. | Zhao, T., et al. 2022. J Med Chem. 65: 4218-4237. PMID: 35084182
- Synthesis and structure-activity relationships of ticlopidine derivatives and analogs as inhibitors of ectonucleotidase CD39. | Bi, C., et al. 2023. Bioorg Chem. 135: 106460. PMID: 37023582
- Pd‐Catalyzed Chemoselective Suzuki‐Miyaura Reactions of 1‐Bromo‐4‐(halomethyl) naphthalene | Pei, M., Liu, P., Ma, X., Liu, Y., & Dai, B. 2018. ChemistrySelect. 3(17): 5002-5004.
- Carbocation Catalyzed Bromination of Alkyl Arenes, a Chemoselective sp3 vs. sp2 C− H functionalization | Ni, S., El Remaily, M. A. E. A. A. A., & Franzén, J. 2018. Advanced Synthesis & Catalysis. 360(21): 4197-4204.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
1-Bromo-4-(bromomethyl)naphthalene, 1 g | sc-297817 | 1 g | $87.00 | |||
1-Bromo-4-(bromomethyl)naphthalene, 5 g | sc-297817A | 5 g | $342.00 |
