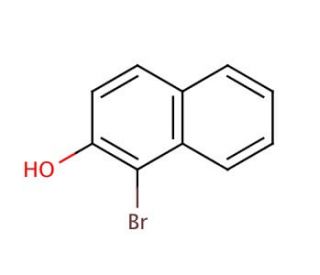
Molecular structure of 1-Bromo-2-naphthol, CAS Number: 573-97-7
1-Bromo-2-naphthol (CAS 573-97-7)
CAS Number:
573-97-7
Molecular Weight:
223.07
Molecular Formula:
C10H7BrO
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
1-Bromo-2-naphthol is a halogenated aromatic compound notable for its role in the synthesis of other chemicals and as a laboratory analytical reagent. While it is soluble in typical organic solvents like ethanol and acetone, it remains insoluble in water. Given its aromatic nature, it partakes in standard reactions like electrophilic substitutions. Widely used in detecting alkaloids, amino acids, and halogens, this compound also aids in producing chemicals, including dyes. 1-Bromo-2-naphthol contributes to the exploration of biochemical pathways.
1-Bromo-2-naphthol (CAS 573-97-7) References
- Synthesis and structure-activity relationship of (1-halo-2-naphthyl) carbamate-based inhibitors of KIAA1363 (NCEH1/AADACL1). | Shreder, KR., et al. 2012. Bioorg Med Chem Lett. 22: 5748-51. PMID: 22877630
- Modular Assembly of Spirocarbocyclic Scaffolds through Pd0 -Catalyzed Intermolecular Dearomatizing [2+2+1] Annulation of Bromonaphthols with Aryl Iodides and Alkynes. | Zuo, Z., et al. 2017. Angew Chem Int Ed Engl. 56: 2767-2771. PMID: 28128533
- A Dearomatization/Debromination Strategy for the [4+1] Spiroannulation of Bromophenols with α,β-Unsaturated Imines. | Ge, Y., et al. 2020. Angew Chem Int Ed Engl. 59: 18985-18989. PMID: 32662085
- Naphthalene tetrachlorides and related compounds. Part 12. Influence of some 1-substituents on the course of chlorination of derivatives of 2-naphthol | David J. Calvert, Peter B. D. de la Mare and Hitomi Suzuki. 1983. J. Chem. Soc., Perkin Trans. 2: 255-260.
- Arenediazonium o‐Benzenedisulfonimides: Some Kinetics of Azo Coupling Reactions with Naphthols | Boga, C., Degani, J., Vecchio, E. D., Fochi, R., Forlani, L., & Todesco, P. E. 2002. European Journal of Organic Chemistry. 2002(22): 3837-3843.
- Pd‐Catalyzed Annulation of β‐Iodovinyl Sulfones with 2‐Halophenols: A General Route for the Synthesis of 3‐Sulfonyl Benzofuran Derivatives | Reddy, R. J., Kumar, J. J., Kumari, A. H., & Krishna, G. R. 2020. Advanced Synthesis & Catalysis. 362(6): 1317-1322.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
1-Bromo-2-naphthol, 25 g | sc-237481 | 25 g | $70.00 | |||
1-Bromo-2-naphthol, 100 g | sc-237481A | 100 g | $228.00 |
