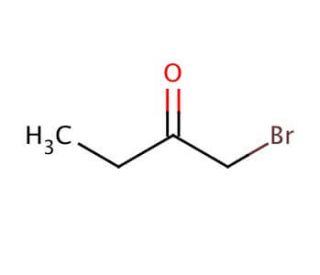
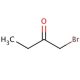
1-Bromo-2-butanone (CAS 816-40-0)
QUICK LINKS
1-Bromo-2-butanone is a compound utilized in organic chemistry research for the study of nucleophilic substitution reactions. It acts as a versatile reagent in the synthesis of various ketones, alcohols, and other organic intermediates. The bromine atom in 1-Bromo-2-butanone provides a reactive site for reaction with nucleophiles, allowing researchers to explore the kinetics and mechanisms of bimolecular substitution processes (SN2 reactions). In addition, this compound is used in the preparation of heterocyclic compounds, which are of interest in the development of new materials and chemicals with specific properties. Studies often focus on the selectivity and yield of reactions involving 1-Bromo-2-butanone, with the aim of optimizing synthetic pathways for efficient production of desired products.
1-Bromo-2-butanone (CAS 816-40-0) References
- Measurements of octanol-air partition coefficients, vapor pressures and vaporization enthalpies of the (E) and (Z) isomers of the 2-ethylhexyl 4-methoxycinnamate as parameters of environmental impact assessment. | Pegoraro, CN., et al. 2015. Chemosphere. 138: 546-52. PMID: 26210018
- Formation of chlorinated breakdown products during degradation of sunscreen agent, 2-ethylhexyl-4-methoxycinnamate in the presence of sodium hypochlorite. | Gackowska, A., et al. 2016. Environ Sci Pollut Res Int. 23: 1886-97. PMID: 26408113
- Degradation of Organic UV filters in Chlorinated Seawater Swimming Pools: Transformation Pathways and Bromoform Formation. | Manasfi, T., et al. 2017. Environ Sci Technol. 51: 13580-13591. PMID: 29110466
- Estimation of physicochemical properties of 2-ethylhexyl-4-methoxycinnamate (EHMC) degradation products and their toxicological evaluation. | Gackowska, A., et al. 2018. Environ Sci Pollut Res Int. 25: 16037-16049. PMID: 29594898
- Two-generation exposure to 2-ethylhexyl 4-methoxycinnamate (EHMC) in Japanese medaka (Oryzias latipes) and its reproduction and endocrine related effects. | Lee, I., et al. 2019. Chemosphere. 228: 478-484. PMID: 31051350
- Analysis of photokinetics of 2'-ethylhexyl-4-methoxycinnamate in sunscreens. | Herzog, B., et al. 2019. Photochem Photobiol Sci. 18: 1773-1781. PMID: 31112187
- Alginate microparticles as carriers for the UV filter 2-ethylhexyl 4-methoxycinnamate: Influence on photostability. | Duarte, J., et al. 2019. Int J Cosmet Sci. 41: 585-593. PMID: 31509264
- Electronic State and Photophysics of 2-Ethylhexyl-4-methoxycinnamate as UV-B Sunscreen under Jet-Cooled Condition. | Muramatsu, S., et al. 2020. J Phys Chem A. 124: 1272-1278. PMID: 31992045
- Effects of Sulfamethoxazole and 2-Ethylhexyl-4-Methoxycinnamate on the Dissimilatory Nitrate Reduction Processes and N2O Release in Sediments in the Yarlung Zangbo River. | Xu, H., et al. 2020. Int J Environ Res Public Health. 17: PMID: 32168922
- Novel green phosphorene sheets to detect tear gas molecules - A DFT insight. | Bhuvaneswari, R., et al. 2020. J Mol Graph Model. 100: 107706. PMID: 32858456
- Effects of 2-ethylhexyl-4-methoxycinnamate (EHMC) on thyroid hormones and genes associated with thyroid, neurotoxic, and nephrotoxic responses in adult and larval zebrafish (Danio rerio). | Chu, S., et al. 2021. Chemosphere. 263: 128176. PMID: 33297144
- Biodegradation of 2-ethylhexyl-4-methoxycinnamate in river sediments and its impact on microbial communities. | Zhang, P., et al. 2021. J Environ Sci (China). 104: 307-316. PMID: 33985734
- Relationship between the ability of sunscreens containing 2-ethylhexyl-4'-methoxycinnamate to protect against UVR-induced inflammation, depletion of epidermal Langerhans (Ia+) cells and suppression of alloactivating capacity of murine skin in vivo. | Walker, SL., et al. 1994. J Photochem Photobiol B. 22: 29-36. PMID: 8151453
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
1-Bromo-2-butanone, 1 g | sc-251479 | 1 g | $114.00 |
