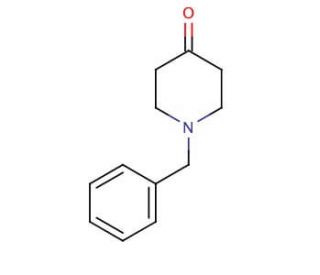
1-Benzyl-4-piperidone (CAS 3612-20-2)
QUICK LINKS
1-Benzyl-4-piperidone is widely used in research applications, particularly in the field of organic synthesis. This compound serves as a key intermediate in the synthesis of various alkaloid derivatives and bioactive compounds. Researchers utilize 1-Benzyl-4-piperidone to explore its reactivity in different chemical reactions, including cyclization processes and nucleophilic additions, which are fundamental for constructing complex molecular architectures. Moreover, it is studied for its potential to form novel compounds with biological activity, aiding in the development of new chemical entities. The studies involving 1-Benzyl-4-piperidone are essential for advancing synthetic methodologies and understanding the chemistry underlying potential new products and chemicals.
1-Benzyl-4-piperidone (CAS 3612-20-2) References
- 4-Hydroxy-1-[2-(4-hydroxyphenoxy)ethyl]-4-(4-methylbenzyl)piperidine: a novel, potent, and selective NR1/2B NMDA receptor antagonist. | Zhou, ZL., et al. 1999. J Med Chem. 42: 2993-3000. PMID: 10425109
- Long-acting fentanyl analogues: synthesis and pharmacology of N-(1-phenylpyrazolyl)-N-(1-phenylalkyl-4-piperidyl)propanamides. | Jagerovic, N., et al. 2002. Bioorg Med Chem. 10: 817-27. PMID: 11814871
- Novel piperidinium and pyridinium agents as water-soluble acetylcholinesterase inhibitors for the reversal of neuromuscular blockade. | Palin, R., et al. 2002. Bioorg Med Chem Lett. 12: 2569-72. PMID: 12182862
- Convenient synthesis of substituted piperidinones from alpha,beta-unsaturated amides: formal synthesis of deplancheine, tacamonine, and paroxetine. | Takasu, K., et al. 2005. J Org Chem. 70: 3957-62. PMID: 15876084
- Synthesis of 4-arylpiperidines from 1-benzyl-4-piperidone: application of the Shapiro reaction and alkenylsilane cross-coupling. | Morrill, C. and Mani, NS. 2007. Org Lett. 9: 1505-8. PMID: 17362023
- Synthesis and potent antileukemic activities of 10-benzyl-9(10H)-acridinones. | Gao, C., et al. 2008. Bioorg Med Chem. 16: 8670-5. PMID: 18713666
- Piperidine derivatives; C-alkylation of l-benzoyl-3-carbethoxy-4-piperidone; synthesis of ethyl 3-ethyl-4-piperidylacetate (d1-ethyl cincholoiponate). | STORK, G. and McELVAIN, SM. 1946. J Am Chem Soc. 68: 1053-7. PMID: 20985619
- Cholinergic and neuroprotective drugs for the treatment of Alzheimer and neuronal vascular diseases. II. Synthesis, biological assessment, and molecular modelling of new tacrine analogues from highly substituted 2-aminopyridine-3-carbonitriles. | Samadi, A., et al. 2011. Bioorg Med Chem. 19: 122-33. PMID: 21163662
- Profiling of illicit fentanyl using UHPLC-MS/MS. | Lurie, IS., et al. 2012. Forensic Sci Int. 220: 191-6. PMID: 22472077
- Design and synthesis of potent antagonists containing rigid spirocyclic privileged structures for the CGRP receptor. | Chaturvedula, PV., et al. 2012. Bioorg Med Chem Lett. 22: 4719-22. PMID: 22732695
- Metabolism of Fentanyl and Acetylfentanyl in Human-Induced Pluripotent Stem Cell-Derived Hepatocytes. | Kanamori, T., et al. 2018. Biol Pharm Bull. 41: 106-114. PMID: 29311471
- Synthesis, Structural and Thermal Studies of 3-(1-Benzyl-1,2,3,6-tetrahydropyridin-4-yl)-5-ethoxy-1H-indole (D2AAK1_3) as Dopamine D₂ Receptor Ligand. | Kondej, M., et al. 2018. Molecules. 23: PMID: 30181442
- Study of the Electrochemical Behavior of N-Substituted-4-Piperidones Curcumin Analogs: A Combined Experimental and Theoretical Approach. | Amalraj, J., et al. 2022. Int J Mol Sci. 23: PMID: 36499370
- Carcinogenic nitrogen compounds. XLVII. Gamma-carbolines and 2,10-diaza-anthracenes isosteric with benzocarbazoles and benzacridines. | Roussel, O., et al. 1965. J Chem Soc Perkin 1. 5458-64. PMID: 4158668
- [Studies on piperidine derivatives. I. Direct synthesis of 4-oxo-1-piperidinecarboxylic acid ester from 1-benzyl-4-piperidone]. | Nakanishi, M. and Arimura, K. 1970. Yakugaku Zasshi. 90: 1324-6. PMID: 5529368
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
1-Benzyl-4-piperidone, 25 g | sc-237473 | 25 g | $31.00 |
