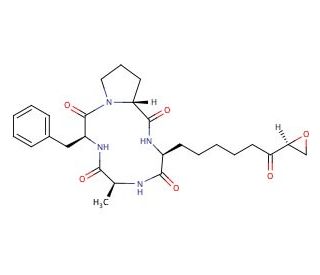
1-Alaninechlamydocin (CAS 141446-96-0)
QUICK LINKS
1-Alaninechlamydocin is a fungal metabolite renowned for its role as an inhibitor of histone deacetylases (HDACs). Its potent ability to reduce overall HDAC activity in HeLa cell lysates is concentration-dependent. Notably, the compound effectively suppresses the proliferation of various cell types, including MIA PaCa-2, PANC-1, and hTERT-HPNE cells, with GI50 values of 5.3, 14, and 2.0 nM, respectively. In particular, 1-Alaninechlamydocin stands out as a remarkable HDAC inhibitor, exerting significant influence on cellular processes. Its impact encompasses inducing G2/M cell cycle arrest and promoting apoptosis specifically in MIA PaCa-2 cells. Furthermore, even at remarkably low concentrations as low as 2 nM, it effectively hampers the activity of SE, further solidifying its potency as an HDAC inhibitor.
1-Alaninechlamydocin (CAS 141446-96-0) References
- A potent HDAC inhibitor, 1-alaninechlamydocin, from a Tolypocladium sp. induces G2/M cell cycle arrest and apoptosis in MIA PaCa-2 cells. | Du, L., et al. 2014. J Nat Prod. 77: 1753-7. PMID: 24999749
- Progress in the Discovery of Macrocyclic Histone Deacetylase Inhibitors for the Treatment of Cancer. | Cheng, K., et al. 2017. Curr Med Chem. 24: 4166-4179. PMID: 28183258
- [1-9-NαC]-crourorb A1 isolated from Croton urucurana latex induces G2/M cell cycle arrest and apoptosis in human hepatocarcinoma cells. | de Matos Cândido-Bacani, P., et al. 2017. Toxicol Lett. 273: 44-54. PMID: 28343894
- Structural Diversity and Biological Activities of Fungal Cyclic Peptides, Excluding Cyclodipeptides. | Wang, X., et al. 2017. Molecules. 22: PMID: 29186926
- Natural Products and Chemical Biology Tools: Alternatives to Target Epigenetic Mechanisms in Cancers. | Lascano, S., et al. 2018. Chem Rec. 18: 1854-1876. PMID: 30537358
- Two New Prenylated Indole Diterpenoids from Tolypocladium sp. and Their Antimicrobial Activities. | Xu, LL., et al. 2019. Chem Biodivers. 16: e1900116. PMID: 30957928
- Chemical genomics reveals histone deacetylases are required for core regulatory transcription. | Gryder, BE., et al. 2019. Nat Commun. 10: 3004. PMID: 31285436
- Cyclic Tetrapeptides from Nature and Design: A Review of Synthetic Methodologies, Structure, and Function. | Sarojini, V., et al. 2019. Chem Rev. 119: 10318-10359. PMID: 31418274
- Epoxide containing molecules: A good or a bad drug design approach. | Gomes, AR., et al. 2020. Eur J Med Chem. 201: 112327. PMID: 32526552
- Sequences of Tolypins, Insecticidal Efrapeptin-Type Peptaibiotics from Species of the Fungal Genus Tolypocladium. | Brückner, H. and Degenkolb, T. 2020. Chem Biodivers. 17: e2000276. PMID: 32573986
- Natural Products Impacting DNA Methyltransferases and Histone Deacetylases. | Akone, SH., et al. 2020. Front Pharmacol. 11: 992. PMID: 32903500
- Denigrins and Dactylpyrroles, Arylpyrrole Alkaloids from a Dactylia sp. Marine Sponge. | Kang, U., et al. 2020. J Nat Prod. 83: 3464-3470. PMID: 33151696
- Cyclic Tetrapeptide HDAC Inhibitors with Improved Plasmodium falciparum Selectivity and Killing Profile. | Collins, JE., et al. 2021. ACS Infect Dis. 7: 2889-2903. PMID: 34491031
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
1-Alaninechlamydocin, 500 µg | sc-391717 | 500 µg | $208.00 |
