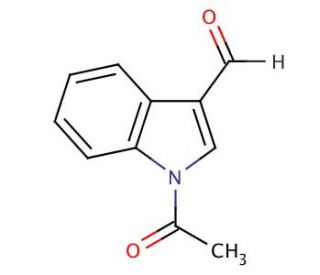

Molecular structure of 1-Acetyl-3-indolecarboxaldehyde, CAS Number: 22948-94-3
1-Acetyl-3-indolecarboxaldehyde (CAS 22948-94-3)
Alternate Names:
N-Acetylindol-3-carboxaldehyde
CAS Number:
22948-94-3
Molecular Weight:
187.19
Molecular Formula:
C11H9NO2
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
1-Acetyl-3-indolecarboxaldehyde functions as a key intermediate in the synthesis of various indole derivatives. 1-Acetyl-3-Indolecarboxaldehyde acts as a versatile building block in the formation of indole-based compounds. Its mechanism of action involves participating in diverse chemical reactions, including condensation, cyclization, and functional group transformations, to yield structurally diverse and biologically active compounds.
1-Acetyl-3-indolecarboxaldehyde (CAS 22948-94-3) References
- Axial-Chiral Biisoquinoline N, N'-Dioxides Bearing Polar Aromatic C-H Bonds as Catalysts in Sakurai-Hosomi-Denmark Allylation. | Reep, C., et al. 2018. Org Lett. 20: 5757-5761. PMID: 30199262
- Synthesis, Characterization, and Crystal Structure Analysis of the First Terdentate [C,N,S] Thiosemicarbazone Complex with a Six-Membered Palladacycle: Influence of Steric Effects on Ring Size | Adriana Amoedo, Luis A. Adrio, José M. Antelo, Javier Martínez, M. Teresa Pereira, Alberto Fernández, José M. Vila. 2006. European Journal of Inorganic Chemistry. 2006: 3016-3021.
- Structure, electronic, spectroscopic and reactivity investigations of pharmacologically active compound 1–acetyl–3–indolecarboxaldehyde – An experimental and theoretical approach | G. Durgadevi a, V. Arjunan b, S. Thirunarayanan a, M.K. Marchewka c, S. Mohan d. 2018. Journal of Molecular Structure. 1164: 57-69.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
1-Acetyl-3-indolecarboxaldehyde, 5 g | sc-255787 | 5 g | $58.00 |
