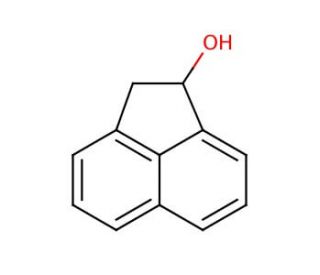
1-Acenaphthenol (CAS 6306-07-6)
QUICK LINKS
1-Acenaphthenol, a synthetic organic compound widely applied in scientific research, emerges as an aromatic hydrocarbon originating from the alkene category, marked by a sole dual connection between two carbon atoms. The configuration of 1-Acenaphthenol encompasses a benzene ring paired with a linked methyl group. In the realm of scientific investigation, 1-Acenaphthenol serves as a reagent across various applications. These encompass the synthesis of alternative organic compounds, gauging chemical reactivity, and delving into reaction mechanisms. Moreover, it operates as a foundational element for generating other aromatic hydrocarbons, notably 2-Acenaphthenol. Within substitution reactions, 1-Acenaphthenol acts as a nucleophile, proficiently displacing halide components in alkyl halides. The process entails formation of a linkage between the alkyl halide′s carbon atom and the hydrogen atom within the 1-Acenaphthenol structure. This tether is subsequently cleaved, liberating the 1-Acenaphthenol molecule while keeping the alkyl group bound to the benzene ring intact.
1-Acenaphthenol (CAS 6306-07-6) References
- Cloning and expression of cDNA encoding hamster liver 3-hydroxyhexobarbital/17beta(3alpha)-hydroxysteroid dehydrogenase 1. | Takenoshita, R., et al. 2001. Chem Biol Interact. 130-132: 863-70. PMID: 11306101
- Oxidation of naphthenoaromatic and methyl-substituted aromatic compounds by naphthalene 1,2-dioxygenase. | Selifonov, SA., et al. 1996. Appl Environ Microbiol. 62: 507-14. PMID: 16535238
- Characterization of the metabolic pathway involved in assimilation of acenaphthene in Acinetobacter sp. strain AGAT-W. | Ghosal, D., et al. 2013. Res Microbiol. 164: 155-63. PMID: 23178176
- New enzymatic assay for the AKR1C enzymes. | Beranič, N., et al. 2013. Chem Biol Interact. 202: 204-9. PMID: 23261716
- Conventional Chiralpak ID vs. capillary Chiralpak ID-3 amylose tris-(3-chlorophenylcarbamate)-based chiral stationary phase columns for the enantioselective HPLC separation of pharmaceutical racemates. | Ahmed, M., et al. 2014. Chirality. 26: 677-82. PMID: 25271972
- Ruthenium complexes as inhibitors of the aldo-keto reductases AKR1C1-1C3. | Traven, K., et al. 2015. Chem Biol Interact. 234: 349-59. PMID: 25446855
- Oxidation of Acenaphthene and Acenaphthylene by Human Cytochrome P450 Enzymes. | Shimada, T., et al. 2015. Chem Res Toxicol. 28: 268-78. PMID: 25642975
- Biodegradation of acenaphthene by Sphingobacterium sp. strain RTSB involving trans-3-carboxy-2-hydroxybenzylidenepyruvic acid as a metabolite. | Mallick, S. 2019. Chemosphere. 219: 748-755. PMID: 30557732
- On the Enantioselective HPLC Separation Ability of Sub-2 µm Columns: Chiralpak® IG-U and ID-U. | Ibrahim, D. and Ghanem, A. 2019. Molecules. 24: PMID: 30986997
- Synthesis and evaluation of AKR1C inhibitory properties of A-ring halogenated oestrone derivatives. | Sinreih, M., et al. 2021. J Enzyme Inhib Med Chem. 36: 1500-1508. PMID: 34227437
- Ruthenium complexes show potent inhibition of AKR1C1, AKR1C2, and AKR1C3 enzymes and anti-proliferative action against chemoresistant ovarian cancer cell line. | Kljun, J., et al. 2022. Front Pharmacol. 13: 920379. PMID: 36034868
- Bacterial oxidation of the polycyclic aromatic hydrocarbons acenaphthene and acenaphthylene. | Schocken, MJ. and Gibson, DT. 1984. Appl Environ Microbiol. 48: 10-6. PMID: 6089663
- Purification and properties of a 3 alpha-hydroxysteroid dehydrogenase of rat liver cytosol and its inhibition by anti-inflammatory drugs. | Penning, TM., et al. 1984. Biochem J. 222: 601-11. PMID: 6435601
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
1-Acenaphthenol, 1 g | sc-237454 | 1 g | $104.00 | |||
1-Acenaphthenol, 5 g | sc-237454A | 5 g | $312.00 |
