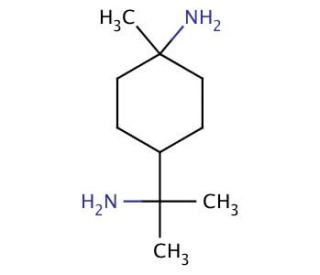
1,8-Diamino-p-menthane (CAS 80-52-4)
QUICK LINKS
1,8-Diamino-p-menthane is a synthetic organic compound characterized by the presence of two amino groups attached to the p-menthane backbone, specifically at the 1 and 8 positions. This structural configuration imparts unique chemical properties, making it an interesting subject for various research applications, particularly in the fields of organic synthesis and material science. The presence of diamino functional groups allows it to act as a bifunctional linker or building block in the construction of complex organic molecules, polymers, or nanomaterials. Its application in research often explores the synthesis of novel compounds with potential for advanced materials, sensors, or catalysts. The specific arrangement of amino groups on the p-menthane skeleton offers avenues for the exploration of stereochemical effects on reactivity and function, enabling studies on chiral chemistry and the synthesis of enantiomerically pure substances. Through its role in the development of new chemical entities, 1,8-Diamino-p-menthane serves as a tool for advancing our understanding of molecular interactions, reaction mechanisms, and the design of materials with desired physical and chemical properties.
1,8-Diamino-p-menthane (CAS 80-52-4) References
- Absorption characteristics of new solvent based on a blend of AMP and 1,8-diamino-p-menthane for CO2 absorption. | Lee, SS., et al. 2012. J Environ Sci (China). 24: 897-902. PMID: 22893968
- Alkylating beta-blockers: activity of isomeric bromoacetyl alprenolol menthanes. | Liptak, A., et al. 1985. J Med Chem. 28: 1699-703. PMID: 2866247
- Affinity labels for beta-adrenoceptors: preparation and properties of alkylating beta-blockers derived from indole. | Pitha, J., et al. 1987. J Med Chem. 30: 612-5. PMID: 2882025
- Beta-adrenoreceptor antagonists with multiple pharmacophores: persistent inhibition of rat heart adenylate cyclase. | Kusiak, JW. and Pitha, J. 1983. J Auton Pharmacol. 3: 195-201. PMID: 6139383
- Binuclear gold(I) complexes with bridging diisocyanides and X-ray structures of [Au(tBuNC)(CN)], [Au2(dmb)(CN)2] and [Au2(tmb)(CN)2] (tmb=2,5-diisocyano-2,5-dimethylhexane; dmb=1,8-diisocyano-p-menthane) | C Chi-Ming, Y Hon-Kay, Wing-Tak Wong, Ting-Fong Lai. 1992. Inorganica Chimica Acta. 197: 177-183.
- Blends of poly(methyl methacrylate) with epoxy resin and an aliphatic amine hardener | CM Gomez, CB Bucknall. 1993. Polymer. 34: 2111-2117.
- Rheological and mechanical properties of poly(butylene terephthalate)-modified epoxy resins | XIA WANG, YIMING LI & YUNZHAO YU. 1997. Journal of Materials Science. 32: 111–114.
- Chemistry, properties and applications of the assembling 1,8-diisocyano-p-menthane, 2,5-dimethyl-2′,5′-diisocyanohexane and 1,3-diisocyanopropane ligands and their coordination polynuclear complexes | PD Harvey. 2001. Coordination Chemistry Reviews. 219–221: 17-52.
- Study of a reactive epoxy–amine resin enabling in situ dissolution of thermoplastic films during resin transfer moulding for toughening composites | M Naffakh, M Dumon, JF Gérard. 2006. Composites Science and Technology. 66: 1376-1384.
- Synthesis and Characterization of Complex Mixtures Consisting of Cyclic and Linear Polyamides from Ethyl Bis-Ketal Galactarates | Cornelia Rosu, et al. 2013. Journal of Macromolecular Science, Part A. 50: 940-952.
- Functionalization of hierarchically porous silica monoliths with polyethyleneimine (PEI) for CO2 adsorption | ingzhong Guo a, Li Ding a, Kazuyoshi Kanamori b, Kazuki Nakanishi b, Hui Yang a. 2017. Microporous and Mesoporous Materials. 245: 51-57.
- Analysis of nitric acid decomposition of epoxy resin network structures for chemical recycling | T Hanaoka, Y Arao, Y Kayaki, S Kuwata. 2021. Polymer Degradation and Stability. 186: 109537.
- Benzotriazole functionalized polydimethylsiloxane for reinforcement water-repellency and corrosion resistance of bio-based waterborne epoxy coatings in salt environment | C Li, Z Xia, H Yan, Q Shi, J Weng. 2022. Corrosion Science. 199: 110150.
- Bio Epoxy Coatings: An Emergent Green Anticorrosive Coating for the Future | and Harikumar Sreehari, Anandhavally Sabarisadhanam Sethulekshmi, Appukuttan Saritha. 2022. 307: 2200004.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
1,8-Diamino-p-menthane, 5 ml | sc-223119 | 5 ml | $43.00 | |||
1,8-Diamino-p-menthane, 100 ml | sc-223119A | 100 ml | $109.00 |
