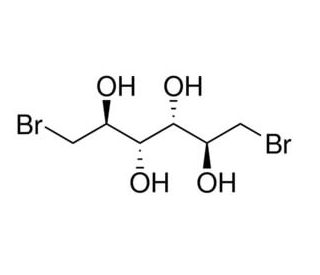
1,6-Dibromo-1,6-dideoxy-D-mannitol (CAS 488-41-5)
QUICK LINKS
1,6-Dibromo-1,6-dideoxy-D-mannitol is a chemically modified sugar alcohol that has been instrumental in various fields of chemical and biochemical research. The introduction of bromine atoms at specific positions in the mannitol molecule makes this compound particularly interesting for studying halogen bonding and its effects on molecular stability and reactivity. This research has implications for understanding and designing halogenated compounds with tailored properties. In the realm of synthetic chemistry, 1,6-Dibromo-1,6-dideoxy-D-mannitol is used to explore reaction mechanisms where bromine acts as a leaving group or participates in electrophilic additions. Researchers employ this compound to investigate the kinetics and thermodynamics of such reactions, providing insights that are crucial for advancing synthetic methodologies. Furthermore, this modified mannitol serves as a building block in the synthesis of more complex molecules. Its structure allows for the creation of various derivatives through substitution reactions, which are studied for their potential as intermediates in organic synthesis. These derivatives can exhibit unique physical and chemical properties useful for a range of industrial applications, from material science to fine chemicals manufacturing. Additionally, 1,6-Dibromo-1,6-dideoxy-D-mannitol is utilized in studies focused on the environmental behavior of brominated organic compounds, particularly their stability, biodegradability, and interactions with natural organic matter. Such research helps in assessing the ecological impact of halogenated compounds, furthering our understanding of their lifecycle and environmental fate.
1,6-Dibromo-1,6-dideoxy-D-mannitol (CAS 488-41-5) References
- Synthesis of 4-substituted phenyl 2,5-anhydro-1,6-dithio-alpha-D-gluco- and -alpha-L-guloseptanosides possessing antithrombotic activity. | Bozó, E., et al. 2000. Carbohydr Res. 329: 25-40. PMID: 11086683
- Synthesis of 4-cyano- and 4-nitrophenyl 2,5-anhydro- 1,6-dithio-alpha-D-gluco- and -alpha-L-guloseptanosides carrying different substituents at C-3 and C-4. | Bozó, E., et al. 2000. Carbohydr Res. 329: 269-86. PMID: 11117311
- The effect of cytostatic hexitol derivatives on haematopoietic stem cells, the L1210 leukaemia and C22LR osteosarcoma. | Fuzy, M., et al. 1975. Eur J Cancer (1965). 11: 169-73. PMID: 165939
- Synthesis of new sugar derivatives having potential anti-tumour activity. XVI. Derivatives of D-mannitol which are differently substituted at C-1 and C-6. | Kuszmann, J. and Vargha, L. 1971. Carbohydr Res. 17: 309-18. PMID: 5150893
- Comparative data on the action mechanism of 1,6-dibromo-1,6-dideoxy-D-mannitol and 1,4-dimethanesulfonyloxy-n-butane. | Institóris, L., et al. 1966. Arzneimittelforschung. 16: 45-50. PMID: 5233532
- Ultrastructural alterations caused by cytostatic sugar-alcohol derivatives. | Lapis, K. and Benedeczky, I. 1968. Cancer Res. 28: 1256-74. PMID: 5665983
- Toxicity, haematological effects and anti-tumour activity of epoxides derived from disubstituted hexitols. Mode of action of mannitol myleran and dibromomannitol. | Elson, LA., et al. 1968. Eur J Cancer (1965). 4: 617-25. PMID: 5717958
- Cross-resistance studies on 1,6-dibromo-dideoxy-D-mannitol(DBM)-resistant Yoshida S.C. sarcoma. | Csányi, E. and Halász, M. 1967. Br J Cancer. 21: 353-7. PMID: 6028086
- Substrate and phospholipid specificity of the purified mannitol permease of Escherichia coli. | Jacobson, GR., et al. 1983. J Cell Biochem. 23: 231-40. PMID: 6427236
- Determination of pharmaceutical compounds containing covalently-bound halogen by means of ion-selective electrodes | F. Rakiás, K. Tóth, E. Pungor. 1980. Analytica Chimica Acta. 121: 93-99.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
1,6-Dibromo-1,6-dideoxy-D-mannitol, 250 mg | sc-213544 | 250 mg | $190.00 | |||
1,6-Dibromo-1,6-dideoxy-D-mannitol, 1 g | sc-213544A | 1 g | $520.00 | |||
1,6-Dibromo-1,6-dideoxy-D-mannitol, 5 g | sc-213544B | 5 g | $2540.00 |
