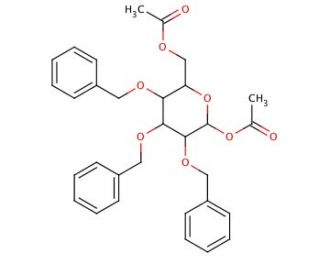
1,6-Di-O-acetyl-2,3,4-tri-O-benzyl-α-D-mannopyranose (CAS 65556-30-1)
QUICK LINKS
1,6-Di-O-acetyl-2,3,4-tri-O-benzyl-α-D-mannopyranose is a chemically modified form of mannose, extensively used in synthetic carbohydrate chemistry for its protective group strategy, enabling controlled reactions crucial for constructing complex glycostructures. This molecule features acetyl groups at the 1 and 6 positions and benzyl groups at the 2, 3, and 4 positions, effectively shielding these reactive hydroxyl groups from undesired side reactions. This selective protection is pivotal for the synthesis of precise glycosidic linkages, which are fundamental in the assembly of sophisticated oligosaccharides and glycoconjugates. In scientific research, this compound serves as a valuable intermediate in the development of mannose-containing polysaccharides, which are significant for studying carbohydrate-protein interactions, particularly those involving lectins, which play crucial roles in cellular recognition and signaling. The specific α-configuration of the glycosidic bond in this derivative is especially important for mimicking the natural orientation of mannose in biological systems, providing insights into the structural requirements for biological activity and interaction. Moreover, the use of 1,6-Di-O-acetyl-2,3,4-tri-O-benzyl-α-D-mannopyranose in glycosylation reactions helps explain the mechanisms of enzyme specificity and catalysis in glycosyl transfer reactions, aiding in the broader understanding of enzymatic processes in glycoscience. This research not only advances the field of carbohydrate chemistry but also contributes to the development of novel synthetic methodologies and materials based on carbohydrate functionalities.
1,6-Di-O-acetyl-2,3,4-tri-O-benzyl-α-D-mannopyranose (CAS 65556-30-1) References
- Stereocontrolled syntheses of alpha-C-mannosyltryptophan and its analogues. | Nishikawa, T., et al. 2005. Org Biomol Chem. 3: 687-700. PMID: 15703809
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
1,6-Di-O-acetyl-2,3,4-tri-O-benzyl-α-D-mannopyranose, 1 g | sc-220571 | 1 g | $320.00 |
