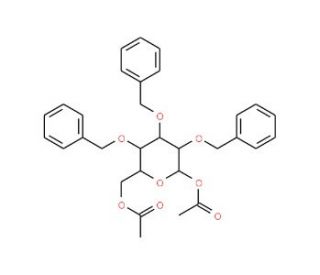
1,6-Di-O-acetyl-2,3,4-tri-O-benzyl-α,β-D-glucopyranose (CAS 59433-13-5)
QUICK LINKS
1,6-Di-O-acetyl-2,3,4-tri-O-benzyl-α,β-D-glucopyranose is a compound that functions as a protecting group in carbohydrate chemistry. It is used to block specific hydroxyl groups in sugar molecules, preventing unwanted reactions at those sites during chemical synthesis. 1,6-Di-O-Acetyl-2,3,4-Tri-O-Benzyl-Α,β-D-Glucopyranose′s mechanism of action involves the acetyl and benzyl groups forming stable covalent bonds with the hydroxyl groups of the sugar molecule, effectively masking them from participating in other chemical reactions. It allows for selective modification of the remaining hydroxyl groups, enabling the synthesis of complex carbohydrate derivatives with precise control over the site of modification. 1,6-Di-O-Acetyl-2,3,4-Tri-O-Benzyl-Α,β-D-Glucopyranose′s role in carbohydrate chemistry is to facilitate the synthesis of structurally diverse sugar derivatives for various purposes, without affecting the structure and properties of the original sugar molecule.
1,6-Di-O-acetyl-2,3,4-tri-O-benzyl-α,β-D-glucopyranose (CAS 59433-13-5) References
- Synthesis of the methyl alpha-glycosides of some isomalto-oligosaccharides specifically deoxygenated at position C-4. | Petráková, E. and Glaudemans, CP. 1995. Carbohydr Res. 279: 133-50. PMID: 8593619
- Synthesis of the methyl alpha-glycosides of some isomalto-oligosaccharides specifically deoxygenated at position C-3. | Petráková, E. and Glaudemans, CP. 1996. Carbohydr Res. 284: 191-205. PMID: 8653719
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
1,6-Di-O-acetyl-2,3,4-tri-O-benzyl-α,β-D-glucopyranose, 250 mg | sc-213542 | 250 mg | $300.00 |
