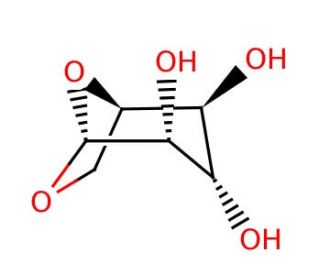
1,6-Anhydro-β-D-glucopyranose (CAS 498-07-7)
QUICK LINKS
1,6-Anhydro-β-D-glucopyranose is a compound of interest in analytical chemistry, particularly in the study of pyrolysis of carbohydrates since it is a major product formed when cellulose is thermally decomposed. Its formation and quantification are for understanding the breakdown mechanisms of biomass and are thus studied in the context of biofuel production and environmental monitoring of biomass burning. In atmospheric chemistry, 1,6-Anhydro-β-D-glucopyranose serves as a tracer for cellulose combustion in airborne particulate matter, aiding in the assessment of contributions from biomass burning to air quality. Researchers also utilize this anhydrosugar as a starting material in synthetic chemistry to produce a variety of glycosidic structures due to its reactive glycosidic bond, which can be exploited to form linkages with other sugars or compounds. Additionally, it is investigated for its potential role in the formation of bio-based polymers, where its anhydride form offers unique reactive properties.
1,6-Anhydro-β-D-glucopyranose (CAS 498-07-7) References
- On hydrogen bonding in 1,6-anhydro-beta-D-glucopyranose (levoglucosan): X-ray and neutron diffraction and DFT study. | Smrcok, L., et al. 2006. Acta Crystallogr B. 62: 912-8. PMID: 16983171
- Stereocontrolled synthesis of lankanolide from 1,6-anhydro-beta-D-glucopyranose (levoglucosan): 1, synthesis of the C-1/7 and C-8/15 segments. | Kochetkov, NK., et al. 1990. Carbohydr Res. 200: 209-25. PMID: 2379206
- 1,6-Anhydro-beta-D-glucopyranose (beta-glucosan), a constituent of human urine. | Dorland, L., et al. 1986. Clin Chim Acta. 159: 11-6. PMID: 3757263
- Molecular motions in solid 1,6-anhydro-β-D-glucopyranose by 1H nuclear magnetic resonance | George W. Smith and F. Shafizadeh. Issue 0, 1971. Journal of the Chemical Society B: Physical Organic. Issue 0: 908-911.
- Konformationsanalyse, XIII1) Umformung von 1,6-Anhydro-β-D-glucopyranose-Ringen in Bootkonformationen durch polare 1,3-diaxiale Wechselwirkungen. Synthese der 2,4-Diamino-1,6-anhydro-2,4-didesoxy-β-D-glucopyranose | . Januar 1976. Chemische Berichte. Volume109, Issue1: Pages 104-111.
- 1H NMR study of 1, 6-Anhydro-β-D-Glucopyranose Tosylated at 2- and/or 4-Position | and Ronan van Rijsbergen, Marc J. O. Anteunis, André De Bruyn. 1982. Bulletin des Sociétés Chimiques Belges. Volume91, Issue4: Pages 297-300.
- Preparation of 2-amino-2,4-dideoxy-D-lyxo-hexopyranose (4-deoxy-D-mannosamine) from 1,6-anhydro-β-D-glucopyranose | Ivan Černý, Tomáš Trnka and Miloslav Černý. 1983,. Collect. Czech. Chem. Commun. 48,: 2386-2394.
- Thioacetolysis Reactions of 1,6-Anhydro-β-D-Glucopyranose Derivatives | Lai-xi Wang, et al. 1990 -. Journal of Carbohydrate Chemistry. Volume 9, Issue 4: Pages 441-450.
- The synthesis of 4-O-oligoethylene glycol derivatives of 1,6-anhydro-β-D-glucopyranose and crown-ethers formed by their intramolecular cyclization | Jindřich Jindřicha, Miloslav Černýb, Tomáš Trnkab and Miloš Buděšínskýa. 1991. Collect. Czech. Chem. Commun.,. 56,: 2950-2963.
- Regioselective acylation of 1,6-anhydro-β-D-glucopyranose catalysed by lipases | C Chon, A Heisler, N Junot, F Levayer… - Tetrahedron: Asymmetry, 1993 - Elsevier. 1993,. Tetrahedron: Asymmetry. Volume 4, Issue 12,: Pages 2441-2444.
- Regioselective acylation of 1,6-anhydro-β-D-manno and galactopyranose catalysed by lipases | N Junot, JC Meslin, C Rabiller - Tetrahedron: Asymmetry, 1995 - Elsevier. 1995,. Tetrahedron: Asymmetry. Volume 6, Issue 6, June: Pages 1387-1392.
- Potentiometric and spectroscopic studies of the binding of copper(II) ions by aminodeoxy derivatives of 1,6-anhydro-β-d-glucopyranose | M Jeżowska-Bojczuk, H Kozłowski, P Decock… - Carbohydrate …, 1992 - Elsevier. 2 September 1992,. Carbohydrate Research. Volume 216,: Pages 453-460.
- Kinetics and mechanism of the acid-catalysed butanolysis of 1,6-anhydro-β-d-glucopyranose | AJJ Straatho, JM Vrolijk, H van Bekkum… - r. 31 December 1988,. Carbohydrate Research. Volume 184,: Pages 163-169.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
1,6-Anhydro-β-D-glucopyranose, 2 g | sc-220561 | 2 g | $141.00 | |||
1,6-Anhydro-β-D-glucopyranose, 5 g | sc-220561A | 5 g | $270.00 | |||
1,6-Anhydro-β-D-glucopyranose, 25 g | sc-220561B | 25 g | $255.00 | |||
1,6-Anhydro-β-D-glucopyranose, 100 g | sc-220561C | 100 g | $525.00 |
