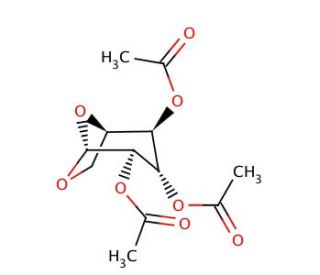
1,6-Anhydro-β-D-glucopyranose 2,3,4-triacetate (CAS 13242-55-2)
QUICK LINKS
1,6-Anhydro-β-D-glucopyranose 2,3,4-triacetate, a modified glucose derivative, has emerged as a valuable tool in glycobiology research, offering insights into carbohydrate chemistry and cellular processes. Its mechanism of action centers around its structural similarity to natural glucose molecules, enabling its use as a substrate analog for enzymes involved in glucose metabolism. Researchers utilize this compound to investigate enzymatic mechanisms and kinetics through biochemical assays and structural studies, shedding light on the complex processes governing carbohydrate metabolism. Furthermore, it serves as a precursor molecule for the synthesis of glycoconjugates and glycomimetics with tailored structures, facilitating the study of glycan-mediated signaling pathways and cellular interactions. This compound has also found applications in carbohydrate-based materials science, where it contributes to the development of biomaterials and drug delivery systems. Additionally, it plays a crucial role in carbohydrate synthesis strategies, enabling the construction of complex glycan structures for glycomic and glycoproteomic analyses. Through its versatile applications, 1,6-Anhydro-β-D-glucopyranose 2,3,4-triacetate continues to advance our understanding of carbohydrate biology and pave the way for innovative research approaches aimed at unraveling the roles of glycans in biological systems.
1,6-Anhydro-β-D-glucopyranose 2,3,4-triacetate (CAS 13242-55-2) References
- A new anhydride of D-glucose: D-glucosan | DIMLER, RJ., et al. 1946. J Am Chem Soc. 68: 1377-80. PMID: 20991009
- A biomimetic synthesis of (+)-biotin from D-glucose. | Ogawa, T., et al. 1977. Carbohydr Res. 57: C31-5. PMID: 902272
- Preparation of Triacetyllevoglucosan. | Coleman, George H., et al. 1944. Industrial & Engineering Chemistry. 36.11: 1040-1041.
- Recent observations on the action of alkali on phenyl glycosides. | MONTGOMERY, EDNA M., et al. 1945. The Journal of Organic Chemistry. 10.3: 194-198.
- Mechanism of the formation of Levoglucosan. | BARDOLPH, MARINUS P. and GEORGE H. COLEMAN. 1950. The Journal of Organic Chemistry. 15.1: 169-173.
- Regioselectively Modified Stereoregular Polysaccharide. 1. Polymerization of 1, 6-Anhydro-3-O-acetyl-2, 4-di-O-benzyl-β-D-glucopyranose and Synthesis of 2, 4-Di-O-benzyl-(1→ 6)-α-D-glucopyranan. | Kobayashi, Kazukiyo, et al. 1979. Macromolecules. 12.6: 1019-1023.
- Microwave pyrolysis of cellulosic materials for the production of anhydrosugars. | Miura, Masakatsu, et al. 2001. Journal of wood science. 47: 502-506.
- Levoglucosan, cellobiose and their acetates as model compounds for the thermally assisted hydrolysis and methylation of cellulose and cellulose acetate. | Schwarzinger, C., et al. 2002. Journal of Analytical and Applied Pyrolysis. 62.2: 179-196.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
1,6-Anhydro-β-D-glucopyranose 2,3,4-triacetate, 1 g | sc-220562 | 1 g | $300.00 |
