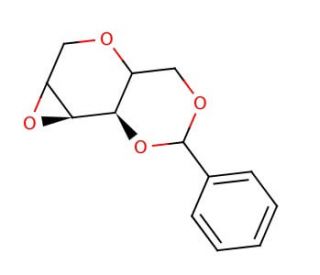
Molecular structure of 1,5:2,3-Dianhydro-4,6-O-benzylidene-D-allitol, CAS Number: 109428-30-0
1,5:2,3-Dianhydro-4,6-O-benzylidene-D-allitol (CAS 109428-30-0)
Application:
1,5:2,3-Dianhydro-4,6-O-benzylidene-D-allitol is useful for synthesis of protected D-Altritol nucleosides
CAS Number:
109428-30-0
Molecular Weight:
234.25
Molecular Formula:
C13H14O4
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
1,5:2,3-Dianhydro-4,6-O-benzylidene-D-allitol is useful for synthesis of protected D-Altritol nucleosides as building blocks for oligonucleotide.
1,5:2,3-Dianhydro-4,6-O-benzylidene-D-allitol (CAS 109428-30-0) References
- Synthesis of D-altritol nucleosides with a 3'-O-tert-butyldimethylsilyl protecting group. | Abramov, M., et al. 2004. Nucleosides Nucleotides Nucleic Acids. 23: 439-55. PMID: 15043165
- Synthesis of altritol nucleoside phosphoramidites for oligonucleotide synthesis. | Abramov, M. and Herdewijn, P. 2007. Curr Protoc Nucleic Acid Chem. Chapter 1: Unit 1.18. PMID: 18428966
- Nucleic acids with a six-membered 'carbohydrate' mimic in the backbone. | Herdewijn, P. 2010. Chem Biodivers. 7: 1-59. PMID: 20087996
- Kinetic analysis of N-alkylaryl carboxamide hexitol nucleotides as substrates for evolved polymerases. | Renders, M., et al. 2019. Nucleic Acids Res. 47: 2160-2168. PMID: 30698800
- Synthesis and antiherpes virus activity of 1,5-anhydrohexitol nucleosides. | Verheggen, I., et al. 1993. J Med Chem. 36: 2033-40. PMID: 8393114
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
1,5:2,3-Dianhydro-4,6-O-benzylidene-D-allitol, 10 mg | sc-220560 | 10 mg | $360.00 |
