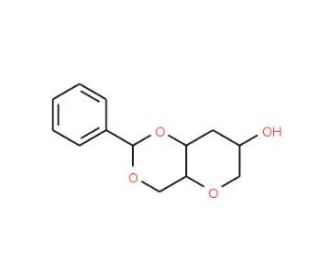
1,5-Anhydro-4,6-O-benzylidene-3-deoxy-D-glucitol (CAS 152613-20-2)
QUICK LINKS
1,5-Anhydro-4,6-O-benzylidene-3-deoxy-D-glucitol is a synthetic carbohydrate derivative that finds its primary utility in carbohydrate chemistry and enzymology research. This compound is particularly useful in studying glycosidic linkage formation and investigating the stereochemistry of carbohydrate-based molecules due to its distinctive structure. As an anhydro sugar, it is often employed as a precursor or intermediate in synthesizing complex oligosaccharides. Its protected hydroxyl groups and benzylidene ring enable researchers to explore selective deprotection and functionalization strategies, crucial for synthesizing carbohydrate derivatives with specific stereochemical configurations. This compound is also used to explain mechanisms of enzymatic reactions involving glycosidic bonds. By acting as a substrate analog, it provides insights into enzyme specificity and catalytic mechanisms, particularly for glycosidases and glycosyltransferases. Additionally, researchers utilize 1,5-anhydro-4,6-O-benzylidene-3-deoxy-D-glucitol to investigate carbohydrate-binding proteins′ recognition patterns. Its stable structure and distinctive benzylidene protection make it a valuable tool in probing carbohydrate-protein interactions, essential for understanding cell surface recognition and signaling. The compound thus contributes significantly to advancing synthetic carbohydrate chemistry, enzymology, and glycobiology by providing a versatile platform for understanding carbohydrate modifications and enzyme mechanisms.
1,5-Anhydro-4,6-O-benzylidene-3-deoxy-D-glucitol (CAS 152613-20-2) References
- Base pairing of anhydrohexitol nucleosides with 2,6-diaminopurine, 5-methylcytosine and uracil asbase moiety. | Boudou, V., et al. 1999. Nucleic Acids Res. 27: 1450-6. PMID: 10037805
- Synthesis of 1,5-Anhydro-d-fructose derivatives and evaluation of their inflammasome inhibitors. | Goto, K., et al. 2018. Bioorg Med Chem. 26: 3763-3772. PMID: 30017113
- Synthesis and antiherpes virus activity of 1,5-anhydrohexitol nucleosides. | Verheggen, I., et al. 1993. J Med Chem. 36: 2033-40. PMID: 8393114
- 5-Substituted pyrimidines with a 1,5-anhydro-2, 3-dideoxy-D-arabino-hexitol moiety at N-1: synthesis, antiviral activity, conformational analysis, and interaction with viral thymidine kinase. | Ostrowski, T., et al. 1998. J Med Chem. 41: 4343-53. PMID: 9784109
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
1,5-Anhydro-4,6-O-benzylidene-3-deoxy-D-glucitol, 10 mg | sc-357485 | 10 mg | $290.00 |
