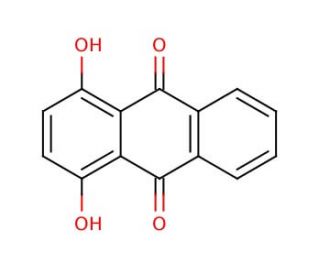
1,4-Dihydroxyanthraquinone (CAS 81-64-1)
QUICK LINKS
1,4-Dihydroxyanthraquinone, a naturally occurring dye found in plants, fungi, and bacteria, boasts a captivating reddish-purple hue, making it a sought-after element for various applications. Its versatility shines through as it finds use in diverse fields like cosmetics, food coloring, and pharmaceuticals. Moreover, 1,4-Dihydroxyanthraquinone proves to be invaluable in the realm of scientific research. One of its pivotal roles lies in shedding light on the impact of light on biological systems, acting as an efficient light absorber within the visible spectrum. Additionally, its high reactivity renders it ideal for measuring the presence of oxidizing agents in cell samples, providing invaluable insights into oxidative stress. Furthermore, 1,4-Dihydroxyanthraquinone unveils its potential in elucidating the workings of enzymes on substrates. Scientists leverage it as a substrate for specific enzymes, allowing them to gain valuable knowledge about enzyme-substrate interactions. However, 1,4-Dihydroxyanthraquinone has yet another intriguing aspect to offer – its ability to act as a photosensitizer. When exposed to light, it sets off a chain reaction, generating reactive oxygen species (ROS). These ROS have the power to engage with cellular components, including proteins and DNA, potentially leading to cellular damage, mutations, or even cell death. In essence, 1,4-Dihydroxyanthraquinone′s vivid color and multifaceted properties make it an essential tool in various scientific investigations, unraveling mysteries that contribute to our understanding of biological systems and cellular behavior.
1,4-Dihydroxyanthraquinone (CAS 81-64-1) References
- Ionic liquid-promoted ring-closure reactions between 1,4-dihydroxyanthraquinone and diamines. | Le, ZG., et al. 2006. Molecules. 11: 464-8. PMID: 17962779
- Study of 1,4-dihydroxyanthraquinone as an acid-base indicator in isopropyl alcohol medium Evaluation of colour-change limits through complementary chromaticity parameters. | Barbosa, J., et al. 1984. Talanta. 31: 279-82. PMID: 18963587
- Theoretical study of molecular structure and vibrational spectra of 1,4-dihydroxyanthraquinone. | Xuan, X., et al. 2011. Spectrochim Acta A Mol Biomol Spectrosc. 79: 1091-8. PMID: 21561800
- Electrospun 1,4-DHAQ-doped cellulose nanofiber films for reusable fluorescence detection of trace Cu2+ and further for Cr3+. | Wang, M., et al. 2012. Environ Sci Technol. 46: 367-73. PMID: 22129160
- Spectroscopic and in silico evaluation of interaction of DNA with six anthraquinone derivatives. | Ghosh, P., et al. 2013. Appl Biochem Biotechnol. 170: 1127-37. PMID: 23645388
- Synthesis of new cytotoxic aminoanthraquinone derivatives via nucleophilic substitution reactions. | Nor, SM., et al. 2013. Molecules. 18: 8046-62. PMID: 23884135
- Combined spectral experiment and theoretical calculation to study the interaction of 1,4-dihydroxyanthraquinone for metal ions in solution. | Yin, C., et al. 2013. Spectrochim Acta A Mol Biomol Spectrosc. 115: 772-7. PMID: 23892118
- Solvent effects on the photophysical properties of poly[1,4-dihydroxyanthraquinoneimine-1,3-bis(phenylene-ester-methylene)tetramethyldisiloxane]. | Dorneanu, PP., et al. 2015. Spectrochim Acta A Mol Biomol Spectrosc. 134: 218-24. PMID: 25014644
- Exploring 1,4-dihydroxyanthraquinone as long-range emissive ratiometric fluorescent probe for signaling Zn(2+)/PO4(3-): Ensemble utilization for live cell imaging. | Sinha, S., et al. 2015. J Photochem Photobiol B. 148: 181-187. PMID: 25956560
- 1,4-dihydroxyanthraquinone electrochemical sensor based on molecularly imprinted polymer using multi-walled carbon nanotubes and multivariate optimization method. | Nezhadali, A., et al. 2016. Talanta. 146: 525-32. PMID: 26695300
- Design, synthesis and biological evaluation of 1,4-dihydroxyanthraquinone derivatives as anticancer agents. | Liu, Y., et al. 2019. Bioorg Med Chem Lett. 29: 1120-1126. PMID: 30846253
- Cyclic synthesis of lignin anthraquinone electrolytes for aqueous redox flow batteries. | Jiao, L., et al. 2023. Int J Biol Macromol. 229: 236-246. PMID: 36572085
- Color additives. Determination of 1,4-dihydroxyanthraquinone in D&C green no. 5 and the former external D&C violet no. 2. | Hoskins, EC. 1971. J Assoc Off Anal Chem. 54: 1270-1. PMID: 5151692
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
1,4-Dihydroxyanthraquinone, 50 g | sc-223049 | 50 g | $56.00 | |||
1,4-Dihydroxyanthraquinone, 250 g | sc-223049A | 250 g | $122.00 | |||
1,4-Dihydroxyanthraquinone, 3 kg | sc-223049B | 3 kg | $918.00 |
