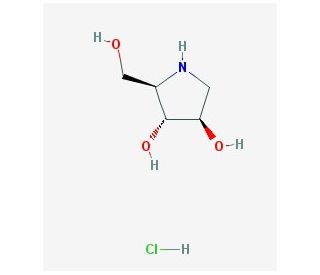
1,4-Dideoxy-1,4-imino-D-arabinitol hydrochloride (CAS 100991-92-2)
See product citations (2)
QUICK LINKS
1,4-Dideoxy-1,4-imino-D-arabinitol Hydrochloride is a glycogen phosphorylase inhibitor. It is also an inhibitor of Glucosidase II alpha, PYGB, PYGL and PYGM. 1,4-Dideoxy-1,4-imino-D-arabinitol hydrochloride, a synthetic compound, has found diverse applications in the realms of science and medicine. As a derivative of 1,4-dideoxy-1,4-imino-D-arabinitol, it serves as a building block for the synthesis of numerous compounds, such as nucleoside analogs, polysaccharides, and peptides. Furthermore, its potential has been extensively explored in various in vitro studies. Notably, it has demonstrated promise in elucidating fundamental cellular processes, including cell proliferation, differentiation, and apoptosis. Moreover, researchers have investigated its potential in unraveling the intricate mechanisms of gene expression and signal transduction pathways.
1,4-Dideoxy-1,4-imino-D-arabinitol hydrochloride (CAS 100991-92-2) References
- Inhibition of glycogenolysis in primary rat hepatocytes by 1, 4-dideoxy-1,4-imino-D-arabinitol. | Andersen, B., et al. 1999. Biochem J. 342 Pt 3: 545-50. PMID: 10477265
- Synthesis of naturally occurring iminosugars from D-fructose by the use of a zinc-mediated fragmentation reaction. | Lauritsen, A. and Madsen, R. 2006. Org Biomol Chem. 4: 2898-905. PMID: 16855738
- Molecular interaction of imino sugars with human alpha-galactosidase: Insight into the mechanism of complex formation and pharmacological chaperone action in Fabry disease. | Sugawara, K., et al. 2009. Mol Genet Metab. 96: 233-8. PMID: 19181556
- Tissue-type plasminogen activator mediates neuroglial coupling in the central nervous system. | An, J., et al. 2014. Neuroscience. 257: 41-8. PMID: 24200922
- Glycogen metabolism protects against metabolic insult to preserve carotid body function during glucose deprivation. | Holmes, AP., et al. 2014. J Physiol. 592: 4493-506. PMID: 25063821
- Single-cell imaging of bioenergetic responses to neuronal excitotoxicity and oxygen and glucose deprivation. | Connolly, NM., et al. 2014. J Neurosci. 34: 10192-205. PMID: 25080581
- Regulatory volume increase in astrocytes exposed to hypertonic medium requires β1 -adrenergic Na(+) /K(+) -ATPase stimulation and glycogenolysis. | Song, D., et al. 2015. J Neurosci Res. 93: 130-9. PMID: 25124094
- Astrocytic β2-adrenergic receptors mediate hippocampal long-term memory consolidation. | Gao, V., et al. 2016. Proc Natl Acad Sci U S A. 113: 8526-31. PMID: 27402767
- Retinal ischemia induces α-SMA-mediated capillary pericyte contraction coincident with perivascular glycogen depletion. | Alarcon-Martinez, L., et al. 2019. Acta Neuropathol Commun. 7: 134. PMID: 31429795
- Phosphorylase Kinase β Represents a Novel Prognostic Biomarker and Inhibits Malignant Phenotypes of Liver Cancer Cell. | Yang, W., et al. 2019. Int J Biol Sci. 15: 2596-2606. PMID: 31754332
- Astrocyte L-Lactate Signaling in the ACC Regulates Visceral Pain Aversive Memory in Rats. | Iqbal, Z., et al. 2022. Cells. 12: PMID: 36611820
- Serotonergic neurons control cortical neuronal intracellular energy dynamics by modulating astrocyte-neuron lactate shuttle. | Natsubori, A., et al. 2023. iScience. 26: 105830. PMID: 36713262
- Sugars with nitrogen in the ring isolated from the leaves of Morus bombycis. | Asano, N., et al. 1994. Carbohydr Res. 253: 235-45. PMID: 8156550
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
1,4-Dideoxy-1,4-imino-D-arabinitol hydrochloride, 10 mg | sc-220553 | 10 mg | $431.00 | |||
1,4-Dideoxy-1,4-imino-D-arabinitol hydrochloride, 25 mg | sc-220553A | 25 mg | $1025.00 |
