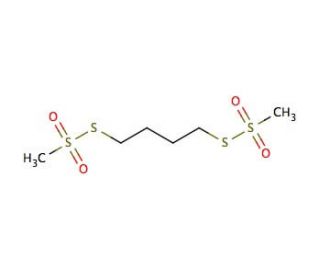
1,4-Butanediyl Bismethanethiosulfonate (CAS 55-99-2)
See product citations (2)
QUICK LINKS
1,4-Butanediyl bismethanethiosulfonate is a chemical compound used as a cleavable crosslinker in protein and peptide research. Researchers utilize this compound for its ability to form reversible disulfide bonds between thiol groups of cysteine residues, which can be useful in studying protein structures and interactions. In proteomics, 1,4-Butanediyl bismethanethiosulfonate is applied to stabilize protein complexes for mass spectrometry analysis, allowing for a more detailed understanding of protein-protein interactions. It is also involved in the reversible modification of cysteine residues in enzymes and other proteins, which aids in elucidating their functions and mechanisms. Additionally, this compound plays a role in the field of materials science, where it is used to modify the surface of gold nanoparticles, providing a reversible linkage for the attachment of thiol-containing molecules.
1,4-Butanediyl Bismethanethiosulfonate (CAS 55-99-2) References
- Evidence of both extra- and intracellular cysteine targets of protein modification for activation of RET kinase. | Akhand, AA., et al. 2002. Biochem Biophys Res Commun. 292: 826-31. PMID: 11944888
- Redox control of catalytic activities of membrane-associated protein tyrosine kinases. | Nakashima, I., et al. 2005. Arch Biochem Biophys. 434: 3-10. PMID: 15629102
- A comparison of electrophysiological properties of the CNGA1, CNGA1tandem and CNGA1cys-free channels. | Mazzolini, M., et al. 2008. Eur Biophys J. 37: 947-59. PMID: 18379773
- Movements of native C505 during channel gating in CNGA1 channels. | Nair, AV., et al. 2009. Eur Biophys J. 38: 465-78. PMID: 19132361
- Conformational rearrangements in the S6 domain and C-linker during gating in CNGA1 channels. | Nair, AV., et al. 2009. Eur Biophys J. 38: 993-1002. PMID: 19488745
- 1,4-butanediyl-bismethanethiosulfonate (BMTS) induces apoptosis through reactive oxygen species-mediated mechanism. | Hossain, K., et al. 2009. J Cell Biochem. 108: 1059-65. PMID: 19830705
- Structural analysis of the extracellular entrance to the serotonin transporter permeation pathway. | Torres-Altoro, MI., et al. 2010. J Biol Chem. 285: 15369-15379. PMID: 20304925
- Control of genetically prescribed protein tyrosine kinase activities by environment-linked redox reactions. | Nakashima, I., et al. 2011. Enzyme Res. 2011: 896567. PMID: 21755044
- The ATPase activity of the P-glycoprotein drug pump is highly activated when the N-terminal and central regions of the nucleotide-binding domains are linked closely together. | Loo, TW., et al. 2012. J Biol Chem. 287: 26806-16. PMID: 22700974
- Identification of the distance between the homologous halves of P-glycoprotein that triggers the high/low ATPase activity switch. | Loo, TW. and Clarke, DM. 2014. J Biol Chem. 289: 8484-92. PMID: 24523403
- Constraining the Lateral Helix of Respiratory Complex I by Cross-linking Does Not Impair Enzyme Activity or Proton Translocation. | Zhu, S. and Vik, SB. 2015. J Biol Chem. 290: 20761-20773. PMID: 26134569
- A short cross-linker activates human P-glycoprotein missing a catalytic carboxylate. | Loo, TW. and Clarke, DM. 2017. Biochem Pharmacol. 145: 27-33. PMID: 28837794
- Activation and closed-state inactivation mechanisms of the human voltage-gated KV4 channel complexes. | Ye, W., et al. 2022. Mol Cell. 82: 2427-2442.e4. PMID: 35597238
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
1,4-Butanediyl Bismethanethiosulfonate, 25 mg | sc-208789 | 25 mg | $330.00 |
