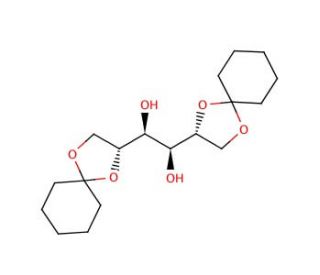
1,2:5,6-Di-O-cyclohexylidene-D-mannitol (CAS 76779-67-4)
QUICK LINKS
1,2:5,6-Di-O-cyclohexylidene-D-mannitol is a compound that functions as a protecting group for hydroxyl groups in organic synthesis. It is used to temporarily mask the hydroxyl groups in carbohydrates, allowing for selective reactions to occur at other functional groups. 1,2:5,6-Di-O-Cyclohexylidene-D-Mannitol acts by forming a stable acetal derivative, which can be easily removed under mild acidic conditions, regenerating the original hydroxyl groups. In this way, 1,2:5,6-Di-O-cyclohexylidene-D-mannitol plays a role in controlling the reactivity of hydroxyl groups in carbohydrate chemistry, enabling the synthesis of complex molecules with specific structural modifications. Its mechanism of action involves the formation of a protective acetal, which prevents unwanted reactions at hydroxyl groups while allowing for targeted chemical transformations at other sites within the molecule. 1,2:5,6-Di-O-Cyclohexylidene-D-Mannitol′s function in organic synthesis involves the precise manipulation of carbohydrate structures, contributing to the development of diverse molecules with tailored properties.
1,2:5,6-Di-O-cyclohexylidene-D-mannitol (CAS 76779-67-4) References
- Organocatalytic Michael Addition to (D)-Mannitol-Derived Enantiopure Nitroalkenes: A Valuable Strategy for the Synthesis of Densely Functionalized Chiral Molecules. | Caruso, L., et al. 2019. Molecules. 24: PMID: 31847419
- Synthesis and Applications of Carbohydrate-Based Organocatalysts. | Wojaczyńska, E., et al. 2021. Molecules. 26: PMID: 34885873
- Rhodium-catalyzed asymmetric hydroformylation of vinylarenes with novel chiral P,N-ligands derived from 1,2:5,6-di-O-cyclohexylidene-d-mannitol | LL Wang, RW Guo, YM Li, ASC Chan - Tetrahedron: Asymmetry, 2005 - Elsevier. 3 October 2005. Tetrahedron: Asymmetry. Volume 16, Issue 19,:, Pages 3198-3204.
- Application of 1,2:5,6-di-O-cyclohexylidene-d-mannitol as the chiral director in Matteson's asymmetric homologation | G Li, GW Kabalka - Journal of organometallic chemistry, 1999 - Elsevier. 5 June 1999,. Journal of Organometallic Chemistry. Volume 581, Issues 1–2,: Pages 66-69.
- Cu-catalyzed enantioselective conjugate addition of diethylzinc to cyclic enones with chiral phosphite ligands derived from 1,2:5,6-di-O-cyclohexylidene-d-mannitol | QL Zhao, LL Wang, FY Kwong, ASC Chan - Tetrahedron: Asymmetry, 2007 - Elsevier. 22 August 2007,. Tetrahedron: Asymmetry. Volume 18, Issue 16,: Pages 1899-1905.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
1,2:5,6-Di-O-cyclohexylidene-D-mannitol, 5 g | sc-222903 | 5 g | $104.00 | |||
1,2:5,6-Di-O-cyclohexylidene-D-mannitol, 25 g | sc-222903A | 25 g | $252.00 |
