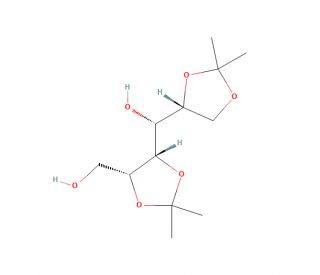
1,2:4,5-Di-O-isopropylidene-D-mannitol (CAS 3969-61-7)
QUICK LINKS
1,2:4,5-Di-O-isopropylidene-D-mannitol, commonly known as DIM, is a versatile compound extensively utilized in organic synthesis and carbohydrate chemistry research. Its mechanism of action primarily involves serving as a protecting group for hydroxyl functionalities in carbohydrates, facilitating selective chemical transformations. Researchers employ DIM as a protecting group strategy to shield specific hydroxyl groups, enabling controlled modifications of sugars during synthetic pathways. This chemical is particularly valuable in carbohydrate chemistry for the synthesis of complex oligosaccharides and glycoconjugates, where precise control over regioselectivity and stereoselectivity is essential. DIM′s ability to temporarily mask hydroxyl groups provides chemists with the flexibility to perform multiple reactions while preserving desired functional groups. Moreover, DIM plays a crucial role in the development of new synthetic methodologies for the construction of diverse carbohydrate structures, enabling investigations into their biological activities and interactions. Its application extends to the synthesis of carbohydrate mimetics, glycomimetics, and glycosidase inhibitors, contributing significantly to the exploration of carbohydrate-based drug discovery and chemical biology research. Overall, 1,2:4,5-Di-O-isopropylidene-D-mannitol is a pivotal tool in carbohydrate chemistry, enabling precise control over synthetic routes and advancing our understanding of carbohydrate-mediated biological processes.
1,2:4,5-Di-O-isopropylidene-D-mannitol (CAS 3969-61-7) References
- Synthesis from D-mannose of 1, 4-dideoxy-1, 4-imino-L-ribitol and of the α-mannosidase inhibitor 1, 4-dideoxy-1, 4-imino-D-talitol. | Fleet, George WJ, et al. 1988. Tetrahedron. 44.9: 2649-2655.
- Chirality induction in cyclopolymerization X. Structural effect of three D-mannitol templates in the cyclocopolymerization of bis (4-vinylbenzoate) with styrene. | Uesaka, Takahiro, et al. 1999. Polymer journal. 31.4: 342-347.
- Chirality Induction in Cyclopolymerization 10. Structural Effect of Three D-Mannitol Templates in the Cyclocopolymerization of Bis (4-vinylbenzoate) with Styrene. | Takahiro, Uesaka, et al. 1999. Polymer Journal. 31.4: 342-347.
- Selective transformation of acetonides to orthoesters: an application of a photoinduced electron transfer process. | Mella, Mariella, et al. 2001. Tetrahedron. 57.3: 555-561.
- Novel mannitol based non-ionic surfactants from biocatalysis: Part two: improved synthesis. | Pinna, Maria Cristina, et al. 2004. Journal of Molecular Catalysis B: Enzymatic. 27.4-6: 233-236.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
1,2:4,5-Di-O-isopropylidene-D-mannitol, 2 g | sc-220542 | 2 g | $360.00 |
