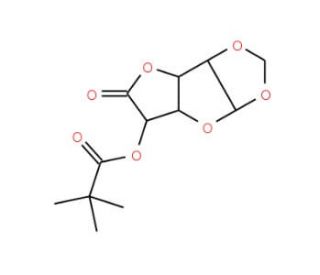
1,2-O-Isopropylidene-α-D-glucofuranosiduronoic Acid 5-o-Pivaloate 6,3-Lactone (CAS 78748-89-7)
QUICK LINKS
1,2-O-Isopropylidene-α-D-glucofuranosiduronoic Acid 5-o-Pivaloate 6,3-Lactone is a chemically modified derivative of D-glucose, extensively utilized in carbohydrate chemistry research for its structural complexity and reactivity. This compound represents a protected form of gluconic acid, where the primary hydroxyl group at the first position is protected with an isopropylidene group, and the carboxylic acid moiety is esterified with pivaloyl (trimethylacetyl) group. Additionally, the lactone ring at the sixth and third positions further enhances its stability and versatility as a synthetic intermediate. Researchers leverage its unique structure in the synthesis of complex carbohydrate derivatives and glycosidic linkages, where precise control over regioselectivity and stereochemistry is crucial. Furthermore, 1,2-O-Isopropylidene-α-D-glucofuranosiduronoic Acid 5-o-Pivaloate 6,3-Lactone finds applications in studying enzyme-catalyzed reactions involved in carbohydrate metabolism and modification. Its use as a substrate or model compound aids in elucidating the mechanisms of glycosyltransferases, glycosidases, and other carbohydrate-modifying enzymes. Moreover, this compound serves as a valuable tool in the development of carbohydrate-based materials with potential applications in areas such as drug delivery, biomaterials, and molecular recognition studies. Overall, its versatility and utility in carbohydrate chemistry research contribute to advancing our understanding of carbohydrate biochemistry and the development of novel carbohydrate-based materials and methodologies.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
1,2-O-Isopropylidene-α-D-glucofuranosiduronoic Acid 5-o-Pivaloate 6,3-Lactone, 250 mg | sc-208750 | 250 mg | $300.00 |
