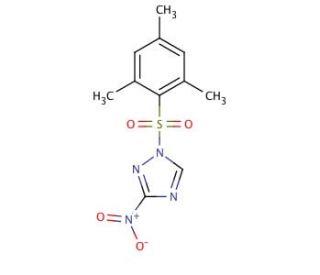
1-(2-Mesitylenesulfonyl)-3-Nitro-1H-1,2,4-Triazole (CAS 74257-00-4)
QUICK LINKS
1-(2-Mesitylenesulfonyl)-3-Nitro-1H-1,2,4-Triazole is a compound utilized in the field of synthetic organic chemistry due to its potential as a versatile reagent in the construction of complex molecules. Its utility is particularly evident in the synthesis of novel heterocyclic compounds, which are essential in materials science for creating advanced polymers and electronics. The mesitylenesulfonyl and nitro groups on the triazole ring offer different points of chemical reactivity, which can be utilized to introduce various functional groups. This dual functionality is leveraged in the design of small molecules for studying enzyme inhibition, where the triazole moiety is a mimic of the transition state in enzymatic reactions. Additionally, this compound is used to produce derivatives with potential applications in the development of sensors and organic semiconductors. In material sciences, researchers explore the energetic properties of nitro-containing triazoles for use in propellants and explosives, where controlled reactivity and stability are paramount. The specificity of its reactivity is also harnessed in photochemistry research, where it can be used to study light-induced electron transfer processes, a fundamental principle behind solar energy conversion.
1-(2-Mesitylenesulfonyl)-3-Nitro-1H-1,2,4-Triazole (CAS 74257-00-4) References
- alpha-Ketocarbonyl peptides: a general approach to reactive resin-bound intermediates in the synthesis of peptide isosteres for protease inhibitor screening on solid support. | Papanikos, A., et al. 2001. J Am Chem Soc. 123: 2176-81. PMID: 11456862
- Synthesis of retro acyl carrier protein (74-65) fragment on a new glycerol based polystyrene support. | Sasikumar, PG., et al. 2003. Protein Pept Lett. 10: 427-33. PMID: 14561130
- The synergy of ChemMatrix resin and pseudoproline building blocks renders RANTES, a complex aggregated chemokine. | García-Martín, F., et al. 2006. Biopolymers. 84: 566-75. PMID: 16810664
- Macrolactamization versus macrolactonization: total synthesis of FK228, the depsipeptide histone deacetylase inhibitor. | Wen, S., et al. 2008. J Org Chem. 73: 9353-61. PMID: 18991384
- Efficient access to peptidyl ketones and peptidyl diketones via C-alkylations and C-acylations of polymer-supported phosphorus ylides followed by hydrolytic and/or oxidative cleavage. | El-Dahshan, A., et al. 2010. Biopolymers. 94: 220-8. PMID: 20225299
- Synthesis and anti-HIV evaluation of new acyclic phosphonate nucleotide analogues and their bis(SATE) derivatives. | Li, H. and Hong, JH. 2010. Nucleosides Nucleotides Nucleic Acids. 29: 581-90. PMID: 20661812
- A spiroligomer α-helix mimic that binds HDM2, penetrates human cells and stabilizes HDM2 in cell culture. | Brown, ZZ., et al. 2012. PLoS One. 7: e45948. PMID: 23094022
- A mild removal of Fmoc group using sodium azide. | Chen, CC., et al. 2014. Amino Acids. 46: 367-74. PMID: 24306456
- AcGly-Phe-Asn(OH) and AcGly-Phe-Asn(NH2) tripeptides selectively affect the proliferation rate of MDA-MB 231 and HuDe cells. | Quassinti, L., et al. 2020. Mol Biol Rep. 47: 4009-4014. PMID: 32277441
- Synthesis and biological evaluation of all possible inosine-mixed cyclic dinucleotides that activate different hSTING variants. | Wang, Z., et al. 2021. Bioorg Med Chem. 29: 115899. PMID: 33285409
- Solid-phase synthesis of curcumin mimics and their anticancer activity against human pancreatic, prostate, and colorectal cancer cell lines. | Romanucci, V., et al. 2021. Bioorg Med Chem. 42: 116249. PMID: 34126286
- Use of the 1-(2-fluorophenyl)-4-methoxypiperidin-4-yl (Fpmp) and related protecting groups in oligoribonucleotide synthesis: stability of internucleotide linkages to aqueous acid. | Capaldi, DC. and Reese, CB. 1994. Nucleic Acids Res. 22: 2209-16. PMID: 8036146
- Automated solid phase synthesis of cyclic oligonucleotides: a further improvement. | De Napoli, L., et al. 1995. Bioorg Med Chem. 3: 1325-9. PMID: 8564398
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
1-(2-Mesitylenesulfonyl)-3-Nitro-1H-1,2,4-Triazole, 1 g | sc-213239 | 1 g | $37.00 | |||
1-(2-Mesitylenesulfonyl)-3-Nitro-1H-1,2,4-Triazole, 5 g | sc-213239A | 5 g | $52.00 | |||
1-(2-Mesitylenesulfonyl)-3-Nitro-1H-1,2,4-Triazole, 25 g | sc-213239B | 25 g | $193.00 | |||
1-(2-Mesitylenesulfonyl)-3-Nitro-1H-1,2,4-Triazole, 100 g | sc-213239C | 100 g | $671.00 | |||
1-(2-Mesitylenesulfonyl)-3-Nitro-1H-1,2,4-Triazole, 250 g | sc-213239D | 250 g | $1618.00 |
