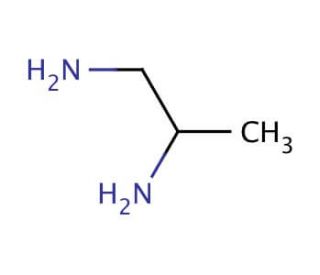
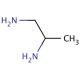
1,2-Diaminopropane (CAS 78-90-0)
QUICK LINKS
1,2-Diaminopropane functions as a diamine in experimental applications. It acts as a crosslinking agent in the synthesis of polymers, particularly in the production of polyamides and epoxy resins. The mechanism of action involves the reaction of 1,2-diaminopropane with carboxylic acid groups, leading to the formation of amide linkages. This process contributes to the enhancement of the mechanical and thermal properties of the resulting polymers. 1,2-diaminopropane can also serve as a building block in the preparation of various organic compounds. Its role in these synthesis pathways involves its ability to participate in nucleophilic substitution reactions, enabling the formation of diverse chemical structures. 1,2-diaminopropane plays a functional role as a crosslinking agent and building block in the synthesis of polymers and organic compounds, contributing to the development of various materials and chemical products.
1,2-Diaminopropane (CAS 78-90-0) References
- Activation and cross-reactivity pattern of a new allergen in adhesive plaster. | Bergendorff, O. and Hansson, C. 2000. Contact Dermatitis. 42: 11-7. PMID: 10644019
- First hexadecavanadate compound: hydrothermal synthesis and characterization of a three-dimensional framework [(Cu(1,2-pn)2)7(V16O38(H2O))2].4H2O. | Lin, BZ. and Liu, SX. 2002. Chem Commun (Camb). 2126-7. PMID: 12362909
- Technological parameters of the ammonolysis of waste 1,2-dichloropropane. | Bartkowiak, M., et al. 2004. J Hazard Mater. 106: 107-14. PMID: 15177099
- Combination of lacunary polyoxometalates and high-nuclear transition metal clusters under hydrothermal conditions. 3. Structure and characterization of [Cu(enMe)2]2{[Cu(enMe)2(H2O)]2[Cu6(enMe)2(B-a-SiW9O34)2]}.4H2O. | Zheng, ST., et al. 2007. Inorg Chem. 46: 4569-74. PMID: 17458958
- Syntheses, spectral, electrochemical and thermal studies of mononuclear manganese(III) complexes with ligands derived from 1,2-propanediamine and 2-hydroxy-3 or 5-methoxybenzaldehyde: self-assembled monolayer formation on nanostructure zinc oxide thin film. | Habibi, MH., et al. 2011. Spectrochim Acta A Mol Biomol Spectrosc. 79: 666-71. PMID: 21536484
- Two tetra-Cd(II)-substituted vanadogermanate frameworks. | Zhou, J., et al. 2014. J Am Chem Soc. 136: 5065-71. PMID: 24641354
- Assemblies of salen-type oxidovanadium(IV) complexes: substituent effects and in vitro protein tyrosine phosphatase inhibition. | Zabierowski, P., et al. 2014. Dalton Trans. 43: 17044-53. PMID: 25303031
- Bis(mandelato)borate: an effective, inexpensive spiroborate anion for chiral resolution. | Wong, LW., et al. 2015. Chem Commun (Camb). 51: 15760-3. PMID: 26369891
- [The Hydrothermal Synthesis, Structure and Spectroscopy Study on (H2dap)6H[V12B16O54(OH)4] · 12H2O (dap = 1, 2-diaminopropane)]. | Guo, ZQ., et al. 2015. Guang Pu Xue Yu Guang Pu Fen Xi. 35: 2625-8. PMID: 26669179
- Substituent changes in the salen ligands of CuIINaI-complexes to induce various structures and catalytic activities towards 2-imidazolines from nitriles and 1,2-diaminopropane. | Li, QQ., et al. 2019. Chem Commun (Camb). 55: 4619-4622. PMID: 30919878
- A series of new hybrid chalcogenogermanates: the rare examples of chalcogenogermanates combined with trivalent vanadium complexes. | Luo, HY., et al. 2019. Dalton Trans. 48: 10907-10914. PMID: 31282904
- From Waste to Schiff Base: Upcycling of Aminolysed Poly(ethylene terephthalate) Product. | Otaibi, AAA., et al. 2022. Polymers (Basel). 14: PMID: 35567031
- Synthesis, characterization, cytotoxic, and DNA binding studies of some platinum (II) complexes of 1,2-diamine and alpha-diimine with 2-pyridinecarboxylate anion. | Paul, AK., et al. 1996. J Inorg Biochem. 61: 179-96. PMID: 9064362
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
1,2-Diaminopropane, 100 g | sc-237669 | 100 g | $31.00 | |||
1,2-Diaminopropane, 500 g | sc-237669A | 500 g | $82.00 |
