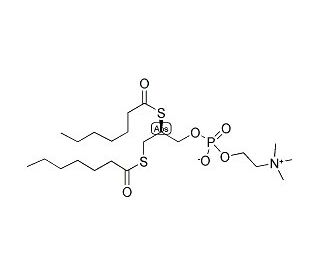
1,2-bis(Heptanoylthio)glycerophosphocholine (CAS 89019-63-6)
QUICK LINKS
1,2-bis(Heptanoylthio)glycerophosphocholine is a synthetic thiol phosphoglyceride derived from D-mannitol which functions as a substrate for phospholipase A2 (PLA2). Hydrolysis of 1,2-bis(Heptanoylthio)glycerophosphocholine by PLA2 produces free thiols, which may be derivitized with dithionitrobenzoic acid to form the chromophore for a spectrophotometric assay of intracellular and membrane-bound PLA2 activity. Release of Arachidonic Acid (sc-200770) from membrane phospholipids mediated by PLA2 is thought to regulate eicosanoid production and marked PLA2 activity is observed in correlation with inflammation states. Inhibitors of PLA2 activity, presenting potential intervention upon the inflammatory role of PLA2, have been identified using this assay design.
1,2-bis(Heptanoylthio)glycerophosphocholine (CAS 89019-63-6) References
- A simple assay for a human serum phospholipase A2 that is associated with high-density lipoproteins. | Petrovic, N., et al. 2001. J Lipid Res. 42: 1706-13. PMID: 11590228
- Inflammatory role of two venom components of yellow jackets (Vespula vulgaris): a mast cell degranulating peptide mastoparan and phospholipase A1. | King, TP., et al. 2003. Int Arch Allergy Immunol. 131: 25-32. PMID: 12759486
- Analysis of human synovial fluid phospholipase A2 on short chain phosphatidylcholine-mixed micelles: development of a spectrophotometric assay suitable for a microtiterplate reader. | Reynolds, LJ., et al. 1992. Anal Biochem. 204: 190-7. PMID: 1514686
- Secreted phospholipase A2 activity in experimental autoimmune encephalomyelitis and multiple sclerosis. | Cunningham, TJ., et al. 2006. J Neuroinflammation. 3: 26. PMID: 16965627
- sPhospholipase A(2) is inhibited by anthocyanidins. | Dreiseitel, A., et al. 2009. J Neural Transm (Vienna). 116: 1071-7. PMID: 19649692
- Multi-targeted activity of maslinic acid as an antimalarial natural compound. | Moneriz, C., et al. 2011. FEBS J. 278: 2951-61. PMID: 21689375
- Effects of novel diarylpentanoid analogues of curcumin on secretory phospholipase A2 , cyclooxygenases, lipo-oxygenase, and microsomal prostaglandin E synthase-1. | Ahmad, W., et al. 2014. Chem Biol Drug Des. 83: 670-81. PMID: 24406103
- Pharmacological evaluation and docking studies of α,β-unsaturated carbonyl based synthetic compounds as inhibitors of secretory phospholipase A₂, cyclooxygenases, lipoxygenase and proinflammatory cytokines. | Bukhari, SN., et al. 2014. Bioorg Med Chem. 22: 4151-61. PMID: 24938495
- Bioactive lysophospholipids generated by hepatic lipase degradation of lipoproteins lead to complement activation via the classical pathway. | Ma, W., et al. 2014. Invest Ophthalmol Vis Sci. 55: 6187-93. PMID: 25205869
- Studies of synthetic chalcone derivatives as potential inhibitors of secretory phospholipase A2, cyclooxygenases, lipoxygenase and pro-inflammatory cytokines. | Jantan, I., et al. 2014. Drug Des Devel Ther. 8: 1405-18. PMID: 25258510
- Synthesis, Molecular Modeling, and Biological Evaluation of Novel 1, 3-Diphenyl-2-propen-1-one Based Pyrazolines as Anti-inflammatory Agents. | Bukhari, SN., et al. 2015. Chem Biol Drug Des. 85: 729-42. PMID: 25328063
- Epididymal phospholipidosis is a possible mechanism for spermatotoxicity induced by the organophosphorus insecticide fenitrothion in rats. | Miyake, M., et al. 2018. Toxicol Lett. 285: 27-33. PMID: 29292090
- Pharmacological appraisal of ligustrazine based cyclohexanone analogs as inhibitors of inflammatory markers. | Alotaibi, NH., et al. 2020. Eur J Pharm Sci. 147: 105299. PMID: 32165315
- Chiral synthesis of a dithiolester analog of phosphatidylcholine as a substrate for the assay of phospholipase A2. | Hendrickson, HS., et al. 1983. J Lipid Res. 24: 1532-7. PMID: 6655369
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
1,2-bis(Heptanoylthio)glycerophosphocholine, 5 mg | sc-201428 | 5 mg | $296.00 |
