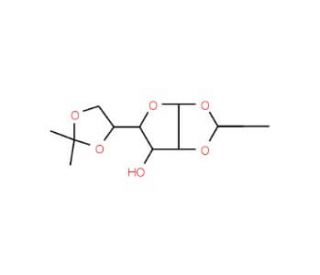
1,2:5,6-Di-O-isopropylidene-α-D-gulofuranose
QUICK LINKS
1,2:5,6-Di-O-isopropylidene-α-D-gulofuranose is a versatile compound extensively utilized in carbohydrate chemistry research due to its unique structural properties and reactivity. Mechanistically, the di-O-isopropylidene protecting groups shield the hydroxyl functional groups, rendering them inert to various chemical reactions. This chemical is commonly employed as a building block in the synthesis of complex carbohydrate derivatives and glycoconjugates. Specifically, it serves as a key intermediate in the preparation of carbohydrate-based materials such as glycan microarrays, carbohydrate-functionalized surfaces, and glycoconjugate vaccines. Additionally, 1,2:5,6-Di-O-isopropylidene-α-D-gulofuranose has been utilized in the development of carbohydrate-based sensors and probes for studying carbohydrate-protein interactions and cellular recognition processes. Its application extends to glycobiology, where it is utilized in investigations concerning glycan biosynthesis pathways, glycosylation reactions, and carbohydrate-mediated biological phenomena. Furthermore, this compound has found utility in the synthesis of glycomimetics, which mimic the structural and functional properties of natural carbohydrates for applications in drug discovery, biomaterials science, and chemical biology. Overall, 1,2:5,6-Di-O-isopropylidene-α-D-gulofuranose plays a crucial role in advancing our understanding of carbohydrate biology and facilitating the development of innovative carbohydrate-based research tools and materials.
1,2:5,6-Di-O-isopropylidene-α-D-gulofuranose References
- A heterogeneous Pd-Bi/C catalyst in the synthesis of L-lyxose and L-ribose from naturally occurring D-sugars. | Fan, A., et al. 2011. Org Biomol Chem. 9: 7720-6. PMID: 21946936
- Synthesis and X-ray studies of novel 3-C-nitromethyl-hexofuranoses. | Turks, M., et al. 2014. Carbohydr Res. 391: 82-8. PMID: 24785391
- p-Toluenesulfonic acid adorned on MCM-41: an efficient and mild catalyst for the regio/chemo-selective hydrolysis of terminal isopropylidene acetals. | Samudrala, RP., et al. 2023. Nucleosides Nucleotides Nucleic Acids. 42: 797-806. PMID: 37039446
- A convenient approach to an advanced intermediate for (+)-lactacystin synthesis | Jozef Gonda, Dávid Maliňák, Margaréta Kováčová, Miroslava Martinková. 2013. Tetrahedron Letters. 54: 6768-6771.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
1,2:5,6-Di-O-isopropylidene-α-D-gulofuranose, 100 mg | sc-213487 | 100 mg | $220.00 |
