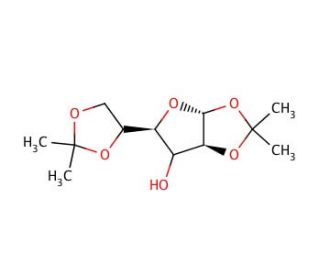
1,2:5,6-Di-O-isopropylidene-α-D-glucofuranose (CAS 582-52-5)
QUICK LINKS
1,2:5,6-Di-O-isopropylidene-α-D-glucofuranose is a chemically modified derivative of glucose, widely utilized in the field of organic synthesis and carbohydrate chemistry for its role as a versatile intermediate. This compound is particularly notable for its protective isopropylidene groups which shield the furanose ring′s reactive hydroxyl groups during chemical reactions, allowing for selective modifications at other sites on the molecule. The presence of these protective groups facilitates the exploration of nucleophilic substitutions and other transformations that are essential in the synthesis of more complex sugar derivatives and oligosaccharides. In research, this compound has been employed to study the kinetics and mechanisms of glycosylation reactions, providing insights into how different conditions affect the reactivity and selectivity of sugar-based synthesis. The furanose form of glucose in this derivative also offers a unique structure for investigating the formation of glycosidic linkages that are less common in nature, thereby expanding the repertoire of synthetic strategies available to chemists. This type of research aids in the development of new methodologies for constructing carbohydrate structures with potential applications in materials science, bioengineering, and the development of high-value fine chemicals.
1,2:5,6-Di-O-isopropylidene-α-D-glucofuranose (CAS 582-52-5) References
- Synthesis of 6-deoxy-L-idose and L-acovenose from 1,2: 5,6-di-O-isopropylidene-alpha-D-glucofuranose. | Hung, SC., et al. 2001. Carbohydr Res. 331: 369-74. PMID: 11398979
- Metallohosts Derived from the Assembly of Sugars around Transition Metals: The Complexation of Alkali Metal Cations. | Piarulli, U., et al. 1997. Inorg Chem. 36: 6127-6133. PMID: 11670249
- Synthesis of D-glucose 3- and 6-[2-(perfluoroalkyl)ethyl] phosphates: a new type of anionic surfactant for biomedical use. | Milius, A., et al. 1992. Carbohydr Res. 229: 323-36. PMID: 1394289
- Large scale isolation of 1,2:3,4-di-O-isopropylidene-α-D-glucoseptanose and 2,3:4,5-di-O-isopropylidene-β-D-glucoseptanose. | Stevens, JD. 2011. Carbohydr Res. 346: 689-90. PMID: 21353207
- On Moffatt dehydration of glucose-derived nitro alcohols. | Lugiņina, J., et al. 2012. Carbohydr Res. 350: 86-9. PMID: 22281179
- Synthesis of 5-amino-5-deoxy-D-galactopyranose and 1,5-dideoxy-1,5-imino-D-galactitol, and their inhibition of alpha- and beta-D-galactosidases. | Legler, G. and Pohl, S. 1986. Carbohydr Res. 155: 119-29. PMID: 3024831
- Synthesis and pharmacological properties of 3-O-derivatives of 1,2,5,6-di-O-isopropylidene-alpha-D-glucofuranose. | Goi, A., et al. 1979. Arzneimittelforschung. 29: 986-90. PMID: 315233
- Synthesis of 9-(6-Deoxy-α-L-Talofuranosyl)-6-Methylpurine and 9-(6-Deoxy-β-D-Allofuranosyl)-6-Methylpurine Nucleosides. | Hassan, AEA., et al. 2020. Curr Protoc Nucleic Acid Chem. 81: e105. PMID: 32255553
- Synthesis of fluorinated and nonfluorinated sugar alkenylphosphonates via highly stereoselective Horner-Wadsworth-Emmons olefination. | Bilska-Markowska, M. and Kaźmierczak, M. 2023. Carbohydr Res. 533: 108941. PMID: 37717483
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
1,2:5,6-Di-O-isopropylidene-α-D-glucofuranose, 25 g | sc-220544 | 25 g | $81.00 |
