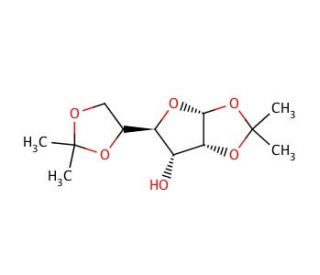
1,2:5,6-Di-O-isopropylidene-α-D-allofuranose (CAS 2595-05-3)
QUICK LINKS
1,2:5,6-Di-O-isopropylidene-α-D-allofuranose has garnered attention in scientific research for its versatile applications, particularly in organic synthesis and carbohydrate chemistry. This compound serves as a valuable building block for the synthesis of complex carbohydrates, glycoconjugates, and related molecules due to its unique structural features. Researchers utilize its functional groups and stereochemistry to access diverse carbohydrate structures and mimic natural glycan motifs. In addition, 1,2:5,6-Di-O-isopropylidene-α-D-allofuranose facilitates the protection and manipulation of hydroxyl groups during carbohydrate synthesis, enabling precise control over regioselectivity and stereochemistry. Its use extends to the development of carbohydrate-based materials, glycomimetics, and glycosylated compounds for applications in chemical biology, materials science, and pharmaceutical research. By incorporating this compound into synthetic pathways, researchers can explore carbohydrate-mediated interactions, develop novel glycan-based probes, and investigate the roles of carbohydrates in biological processes such as cell adhesion, signaling, and immune response. Furthermore, its compatibility with synthetic methodologies allows for the efficient assembly of structurally diverse carbohydrate libraries, contributing to the advancement of glycobiology and the discovery of carbohydrate-based agents. Overall, 1,2:5,6-Di-O-isopropylidene-α-D-allofuranose serves as a valuable tool for researchers in elucidating the structure-function relationships of carbohydrates and designing innovative glycan-based molecules for various research applications.
1,2:5,6-Di-O-isopropylidene-α-D-allofuranose (CAS 2595-05-3) References
- Diastereo- and enantioselective synthesis of alpha,beta-disubstituted gamma-nitro methyl sulfonates. | Enders, D., et al. 2001. Chem Commun (Camb). 2498-9. PMID: 12240033
- A stereoselective asymmetric synthesis of antibiotic (-)-fumagillol using claisen rearrangement and intramolecular ester enolate alkylation as key steps. | Kim, D., et al. 2005. Arch Pharm Res. 28: 129-41. PMID: 15789740
- Short synthesis of octosyl nucleosides. | Knapp, S., et al. 2006. Org Lett. 8: 1335-7. PMID: 16562885
- Influence of the stereochemistry of sugars on the selectivity of formation of carbohydrate-derived cyclopentadienyl and indenyl ligands. | Laï, R., et al. 2006. Dalton Trans. 3478-84. PMID: 16832498
- Synthesis and cytostatic activity of purine nucleosides derivatives of allofuranose. | Besada, P., et al. 2010. Eur J Med Chem. 45: 6114-9. PMID: 20934790
- Cu-Al mixed oxide-catalysed multi-component synthesis of gluco- and allofuranose-linked 1,2,3-triazole derivatives. | Corona-Sánchez, R., et al. 2020. R Soc Open Sci. 7: 200290. PMID: 32874626
- A practical and scalable synthesis of several base modified 3'-O-methyl ribonucleosides. | Rajendra Prasad, S., et al. 2023. Carbohydr Res. 534: 108981. PMID: 37992558
- Synthesis of methyl 2-O-allyl-(and 3-O-allyl)-5-O-benzyl-beta-D- ribofuranoside. | Desai, T., et al. 1996. Carbohydr Res. 280: 209-21. PMID: 8593636
- The synthesis of several octose derivatives related to octosyl acids A and B. | Anzai, Kentaro and Tsuneo Saita. 1977. Bulletin of the Chemical Society of Japan. 50.1: 169-174.
- Evidence on the structure of coyolosa. Synthesis of 6, 6′-ether linked hexoses. | Haines, Alan H. 2004. Tetrahedron letters. 45.4: 835-837.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
1,2:5,6-Di-O-isopropylidene-α-D-allofuranose, 1 g | sc-220543 | 1 g | $140.00 | |||
1,2:5,6-Di-O-isopropylidene-α-D-allofuranose, 10 g | sc-220543A | 10 g | $170.00 | |||
1,2:5,6-Di-O-isopropylidene-α-D-allofuranose, 50 g | sc-220543B | 50 g | $380.00 | |||
1,2:5,6-Di-O-isopropylidene-α-D-allofuranose, 100 g | sc-220543C | 100 g | $540.00 |
