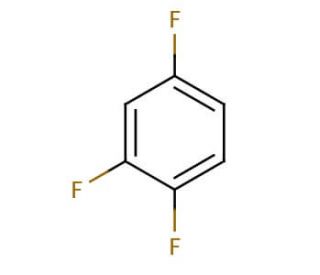
Molecular structure of 1,2,4-Trifluorobenzene, CAS Number: 367-23-7
1,2,4-Trifluorobenzene (CAS 367-23-7)
CAS Number:
367-23-7
Molecular Weight:
132.08
Molecular Formula:
C6H3F3
Supplemental Information:
This is classified as a Dangerous Good for transport and may be subject to additional shipping charges.
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
1,2,4-Trifluorobenzene (TFB) serves as an aromatic organic compound that finds utility as a solvent, catalyst, and reagent across a range of laboratory experiments. The mechanism underlying the action of 1,2,4-trifluorobenzene entails the formation of a complex between the 1,2,4-Trifluorobenzene molecule and the substrate molecule. Subsequently, this complex encounters an electrophile, triggering the establishment of a covalent bond between the two molecules. As the 1,2,4-Trifluorobenzene molecule progresses through a series of subsequent reactions, it ultimately yields the desired product, thus fulfilling its role in the chemical transformation at hand.
1,2,4-Trifluorobenzene (CAS 367-23-7) References
- The effects of volatile aromatic anesthetics on voltage-gated Na+ channels expressed in Xenopus oocytes. | Horishita, T., et al. 2008. Anesth Analg. 107: 1579-86. PMID: 18931215
- Inhibition of human alpha4beta2 neuronal nicotinic acetylcholine receptors by volatile aromatic anesthetics depends on drug hydrophobicity. | Solt, K., et al. 2010. Anesth Analg. 110: 455-60. PMID: 19917625
- Nickel, manganese, cobalt, and iron-catalyzed deprotonative arene dimerization. | Truong, T., et al. 2010. Org Lett. 12: 1200-3. PMID: 20192197
- Ru-Catalyzed C-H Arylation of Fluoroarenes with Aryl Halides. | Simonetti, M., et al. 2016. J Am Chem Soc. 138: 3596-606. PMID: 26942551
- Pyridine-enabled copper-promoted cross dehydrogenative coupling of C(sp2)-H and unactivated C(sp3)-H bonds. | Wu, X., et al. 2015. Chem Sci. 6: 5978-5983. PMID: 29861919
- Non-targeted GC/MS analysis of exhaled breath samples: Exploring human biomarkers of exogenous exposure and endogenous response from professional firefighting activity. | Geer Wallace, MA., et al. 2019. J Toxicol Environ Health A. 82: 244-260. PMID: 30907277
- Predictive Models for the Binary Diffusion Coefficient at Infinite Dilution in Polar and Nonpolar Fluids. | Aniceto, JPS., et al. 2021. Materials (Basel). 14: PMID: 33498723
- Pilot Study on Exhaled Breath Analysis for a Healthy Adult Population in Hawaii. | Yamanaka, HR., et al. 2021. Molecules. 26: PMID: 34207244
- Fluoromaticity: The Molecular Orbital Contributions of Fluorine Substituents to the π-Systems of Aromatic Rings. | Fuhrer, TJ., et al. 2021. ACS Omega. 6: 32607-32617. PMID: 34901609
- Multi-Strategy Assessment of Different Uses of QSAR under REACH Analysis of Alternatives to Advance Information Transparency. | Chinen, K. and Malloy, T. 2022. Int J Environ Res Public Health. 19: PMID: 35410019
- Microwell Fluoride Screen for Chemical, Enzymatic, and Cellular Reactions Reveals Latent Microbial Defluorination Capacity for -CF3 Groups. | Bygd, MD., et al. 2022. Appl Environ Microbiol. 88: e0028822. PMID: 35435713
- Regioselective Magnesiations of Fluorinated Arenes and Heteroarenes Using Magnesium-bis-Diisopropylamide (MBDA) in Hydrocarbons. | Hess, A., et al. 2022. Angew Chem Int Ed Engl. 61: e202206176. PMID: 35575962
- Fluoro-recognition: New in vivo fluorescent assay for toluene dioxygenase probing induction by and metabolism of polyfluorinated compounds. | Aukema, KG., et al. 2022. Environ Microbiol. 24: 5202-5216. PMID: 36054238
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
1,2,4-Trifluorobenzene, 5 g | sc-222889 | 5 g | $44.00 | |||
1,2,4-Trifluorobenzene, 25 g | sc-222889A | 25 g | $152.00 |
