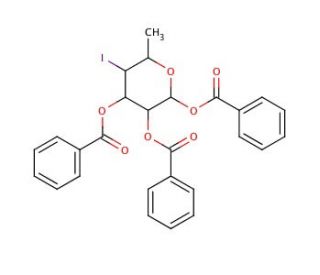
1,2,3-Tri-O-benzoyl-4,6-dideoxy-4-iodo-α-L-glucopyranose (CAS 132867-78-8)
QUICK LINKS
1,2,3-Tri-O-benzoyl-4,6-dideoxy-4-iodo-α-L-glucopyranose serves as a valuable chemical tool in carbohydrate chemistry research, particularly in the synthesis and modification of complex oligosaccharides. The chemical mechanism involves the selective protection of hydroxyl groups on the glucose ring with benzoyl groups, along with the introduction of an iodine atom at the 4-position and the removal of the 6-hydroxyl group, facilitating subsequent glycosylation reactions and the assembly of diverse carbohydrate structures. Researchers utilize this compound to investigate the structural and functional roles of iodinated carbohydrates in various biological processes, including molecular recognition, cellular signaling, and microbial pathogenesis. Additionally, 1,2,3-Tri-O-benzoyl-4,6-dideoxy-4-iodo-α-L-glucopyranose serves as a precursor for the synthesis of radiolabeled carbohydrates for applications in positron emission tomography (PET) imaging and radiotherapy. Its versatility extends to the development of carbohydrate-based materials, such as glycoconjugates, glycomaterials, and carbohydrate-modified nanoparticles, for applications in drug delivery, biomaterials science, and molecular diagnostics. Overall, this chemical plays a crucial role in advancing our understanding of iodinated carbohydrate biology and its implications for various research disciplines, ranging from glycobiology to nanomedicine.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
1,2,3-Tri-O-benzoyl-4,6-dideoxy-4-iodo-α-L-glucopyranose, 25 mg | sc-213458 | 25 mg | $300.00 |
