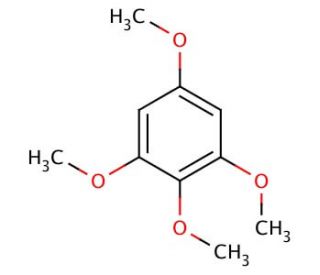
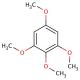
1,2,3,5-Tetramethoxybenzene (CAS 5333-45-9)
QUICK LINKS
1,2,3,5-Tetramethoxybenzene (TMB), characterized by its aromatic hydrocarbon structure, is widely acknowledged for its utility across multiple domains of chemical research. This compound is particularly valuable as a versatile intermediate in the synthesis of complex organic molecules. Its unique properties enable the efficient facilitation of diverse synthetic pathways, contributing significantly to the creation of various high-value chemicals. 1,2,3,5-Tetramethoxybenzene′s efficacy extends to its role in the synthesis of sophisticated dyes, enhancing the palette of colors achievable in industrial applications. Moreover, 1,2,3,5-Tetramethoxybenzene is employed in specialized analytical chemistry techniques for the identification of aldehyde and ketone functional groups, where it acts through a mechanism that involves the stabilization of intermediate complexes, allowing for precise detection and analysis. This functionality underscores its importance in advancing both the methodology and accuracy of chemical analysis in research applications.
1,2,3,5-Tetramethoxybenzene (CAS 5333-45-9) References
- Redox chemistry in laccase-catalyzed oxidation of N-hydroxy compounds. | Xu, F., et al. 2000. Appl Environ Microbiol. 66: 2052-6. PMID: 10788380
- Thermally assisted hydrolysis and methylation of purified tannins from plants. | Nierop, KG., et al. 2005. Anal Chem. 77: 5604-14. PMID: 16131072
- The structure, methyl rotation reflected in inelastic and quasielastic neutron scattering and vibrational spectra of 1,2,3,5-tetramethoxybenzene and its 2:1 complex with 1,2,4,5-tetracyanobenzene. | Pawlukojć, A., et al. 2008. J Chem Phys. 129: 154506. PMID: 19045208
- Direct functionalization of (un)protected tetrahydroisoquinoline and isochroman under iron and copper catalysis: two metals, two mechanisms. | Ghobrial, M., et al. 2011. J Org Chem. 76: 8781-93. PMID: 21902275
- Iron-catalyzed rearrangements and cycloaddition reactions of 2H-chromenes. | Luan, Y., et al. 2011. Org Lett. 13: 6480-3. PMID: 22098535
- Evaluation of bezafibrate, gemfibrozil, indomethacin, sulfamethoxazole, and diclofenac removal by ligninolytic enzymes. | Camarillo Ravelo, D., et al. 2020. Prep Biochem Biotechnol. 50: 592-597. PMID: 32003284
- Total Synthesis of Naturally Occurring 5,7,8-Trioxygenated Homoisoflavonoids. | Kwon, S., et al. 2020. ACS Omega. 5: 11043-11057. PMID: 32455225
- Photocatalyst-Free, Visible-Light-Mediated C(sp3)-H Arylation of Amides via a Solvent-Caged EDA Complex. | Kaur, J., et al. 2021. Org Lett. 23: 2002-2006. PMID: 33596084
- A plant host, Nicotiana benthamiana, enables the production and study of fungal lignin-degrading enzymes. | Khlystov, NA., et al. 2021. Commun Biol. 4: 1027. PMID: 34471192
- De novo synthesis of 4,5-dimethoxycatechol and 2, 5-dimethoxyhydroquinone by the brown rot fungus Gloeophyllum trabeum. | Paszczynski, A., et al. 1999. Appl Environ Microbiol. 65: 674-9. PMID: 9925599
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
1,2,3,5-Tetramethoxybenzene, 1 g | sc-222880 | 1 g | $120.00 | |||
1,2,3,5-Tetramethoxybenzene, 2 g | sc-222880B | 2 g | $200.00 | |||
1,2,3,5-Tetramethoxybenzene, 5 g | sc-222880A | 5 g | $400.00 | |||
1,2,3,5-Tetramethoxybenzene, 10 g | sc-222880C | 10 g | $600.00 | |||
1,2,3,5-Tetramethoxybenzene, 25 g | sc-222880D | 25 g | $1000.00 |
