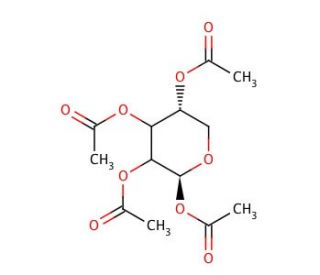
1,2,3,4-Tetra-O-acetyl-D-xylopyranose (CAS 62446-93-9)
QUICK LINKS
1,2,3,4-Tetra-O-acetyl-D-xylopyranose is a carbohydrate derivative extensively utilized in glycobiology research due to its structural resemblance to xylose-containing glycans found in nature. This compound serves as a valuable substrate for enzymatic and chemical glycosylation reactions, facilitating the synthesis of complex oligosaccharides and glycoconjugates. Researchers employ it as a building block in the assembly of structurally diverse carbohydrate structures for investigating carbohydrate-mediated interactions and biological processes. Furthermore, 1,2,3,4-Tetra-O-acetyl-D-xylopyranose is a key component in the development of glycan microarrays, which enable high-throughput screening of carbohydrate-protein interactions and glycan-binding specificities. Its utilization extends to the study of glycan biosynthesis pathways and glycosylation events in cellular processes such as cell adhesion, signaling, and immune response. Additionally, this compound finds application in the synthesis of glycosylated natural products and glycomimetics for drug discovery and development. Overall, 1,2,3,4-Tetra-O-acetyl-D-xylopyranose plays a crucial role in advancing our understanding of glycan diversity, function, and their implications in various biological contexts.
1,2,3,4-Tetra-O-acetyl-D-xylopyranose (CAS 62446-93-9) References
- Solid-phase synthesis of O-linked glycopeptide analogues of enkephalin. | Mitchell, SA., et al. 2001. J Org Chem. 66: 2327-42. PMID: 11281773
- A facile preparation of peracylated alpha-aldopyranosyl chlorides with thionyl chloride and tin tetrachloride. | Wang, Q., et al. 2008. Carbohydr Res. 343: 2989-91. PMID: 18834971
- Nucleic acids with a six-membered 'carbohydrate' mimic in the backbone. | Herdewijn, P. 2010. Chem Biodivers. 7: 1-59. PMID: 20087996
- Synthesis and antiproliferative activity of derivatives of the phyllanthusmin class of arylnaphthalene lignan lactones. | Woodard, JL., et al. 2018. Bioorg Med Chem. 26: 2354-2364. PMID: 29656990
- Triterpenoids and Their Glycosides from Glinus Oppositifolius with Antifungal Activities against Microsporum Gypseum and Trichophyton Rubrum. | Zhang, D., et al. 2019. Molecules. 24: PMID: 31212847
- Synthesis and property of alkyl dioxyethyl α-D-xyloside. | Wu, X., et al. 2020. J Mol Liq. 315: 113770. PMID: 32834260
- Synthesis and cytotoxic analysis of thiolated xylose derivatives decorated on gold nanoparticles. | Shiue, A., et al. 2020. Biotechnol Rep (Amst). 28: e00549. PMID: 33240795
- Xyloside Derivatives as Molecular Tools to Selectively Inhibit Heparan Sulfate and Chondroitin Sulfate Proteoglycan Biosynthesis. | Mencio, C., et al. 2022. Methods Mol Biol. 2303: 753-764. PMID: 34626420
- Synthesis, Antimicrobial and Mutagenic Activity of a New Class of d-Xylopyranosides. | Sikora, K., et al. 2023. Antibiotics (Basel). 12: PMID: 37237791
- Synthesis of a Highly Potent Antitumor Saponin OSW-1 and its Analogues | acek W. Morzycki & Agnieszka Wojtkielewicz. 2005. Phytochemistry Reviews. 4: 259–277.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
1,2,3,4-Tetra-O-acetyl-D-xylopyranose, 1 g | sc-213461 | 1 g | $320.00 |
