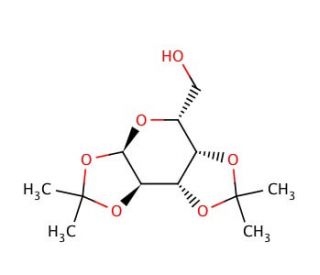
1,2:3,4-Di-O-isopropylidene-α-D-galactopyranose (CAS 4064-06-6)
QUICK LINKS
1,2:3,4-Di-O-isopropylidene-α-D-galactopyranose is a chemical compound widely utilized in carbohydrate chemistry research due to its unique structure and reactivity. This compound serves as a protecting group for the hydroxyl groups on the α-D-galactopyranose moiety, allowing for selective manipulation of specific hydroxyl groups in carbohydrate synthesis. One of its primary research applications is in the synthesis of complex carbohydrates and glycoconjugates, where precise control over the regioselectivity and stereoselectivity of glycosylation reactions is essential. By temporarily masking certain hydroxyl groups, 1,2:3,4-Di-O-isopropylidene-α-D-galactopyranose enables chemists to perform selective reactions on unprotected hydroxyl groups, facilitating the construction of structurally diverse carbohydrate molecules. Additionally, this compound has been employed in the development of carbohydrate-based materials, such as dendrimers and glycopolymer conjugates, for various biomedical and nanotechnology applications. Its versatile reactivity and compatibility with a wide range of synthetic methodologies make it a valuable tool in carbohydrate chemistry research, enabling the synthesis of complex carbohydrate structures with precise control over functional group manipulation. Furthermore, studies continue to explore novel synthetic strategies and applications for 1,2:3,4-Di-O-isopropylidene-α-D-galactopyranose in advancing our understanding of carbohydrate biology and developing new materials with potential biomedical and technological significance.
1,2:3,4-Di-O-isopropylidene-α-D-galactopyranose (CAS 4064-06-6) References
- Syntheses and reactions of 5-O-acetyl-1,2-anhydro-3-O-benzyl-alpha-D-ribofuranose and beta-D-lyxofuranose, 5-O-acetyl-1,2-anhydro-3,6-di-O-benzyl- and 1,2-anhydro-5,6-di-O-benzoyl-3-O-benzyl-beta-D-mannofuranose, and 6-O-acetyl-1,2-anhydro-3,4-di-O-benzyl-alpha-D-glucopyranose and -beta-D-talopyranose. | Ning, J. and Kong, F. 2001. Carbohydr Res. 330: 165-75. PMID: 11217969
- A study of the donor properties of sialyl phosphites having an auxiliary 3-(S)-phenylseleno group. | Ercegovic, T., et al. 2001. Carbohydr Res. 331: 255-63. PMID: 11383895
- Conformation of the galactose ring adopted in solution and in crystalline form as determined by experimental and DFT 1H NMR and single-crystal X-ray analysis. | Roslund, MU., et al. 2004. J Org Chem. 69: 18-25. PMID: 14703374
- Novel perfluoroalkylated derivatives of D-galactopyranose and xylitol for biomedical uses. Hemocompatibility and effect on perfluorocarbon emulsions. | Církva, V., et al. 2004. Carbohydr Res. 339: 2177-85. PMID: 15337445
- Chiral O-(Z-alpha-aminoacyl) sugars: convenient building blocks for glycopeptide libraries. | Katritzky, AR., et al. 2007. Bioconjug Chem. 18: 994-8. PMID: 17441683
- Glycosylated zinc(II) phthalocyanines as efficient photosensitisers for photodynamic therapy. Synthesis, photophysical properties and in vitro photodynamic activity. | Choi, CF., et al. 2008. Org Biomol Chem. 6: 2173-81. PMID: 18528579
- N-Substituted 5-amino-1-hydroxymethyl-cyclopentanetriols: A new family of activity promotors for a GM1-gangliosidosis related human lysosomal β-galactosidase mutant. | Schalli, M., et al. 2017. Carbohydr Res. 443-444: 15-22. PMID: 28319682
- Stereoselective sialylation with O-trifluoroacetylated thiosialosides: hydrogen bonding involved? | Podvalnyy, NM., et al. 2017. Carbohydr Res. 451: 12-28. PMID: 28934626
- Synthesis, conformational analysis, and the glycosidic coupling reaction of substituted 2,7-dioxabicyclo[4.1.0]heptanes: 1,2-anhydro-3,4-di-O-benzyl-beta-L- and beta-D-rhamnopyranoses. | Chen, Q., et al. 1993. Carbohydr Res. 240: 107-17. PMID: 8458005
- Photochemical conversion of sugar dimethylthiocarbamates into deoxy sugars. | Bell, H., et al. 1977. Carbohydr Res. 58: 109-24. PMID: 912673
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
1,2:3,4-Di-O-isopropylidene-α-D-galactopyranose, 5 g | sc-220539 | 5 g | $135.00 |
