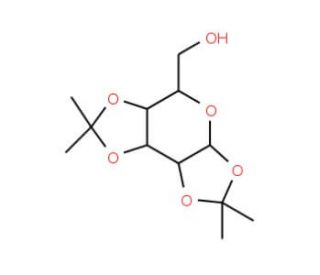
1,2:3,4-Di-O-isopropylidene-α-D-galacto-hexadialdo-1,5-pyranose (CAS 4933-77-1)
QUICK LINKS
1,2:3,4-Di-O-isopropylidene-α-D-galacto-hexadialdo-1,5-pyranose is extensively utilized in synthetic carbohydrate chemistry for its role in constructing complex sugar structures. This chemical acts as a protected form of galactose, where the isopropylidene groups guard the vicinal diol moieties, thus stabilizing the sugar and making it resistant to unwanted reactions during synthetic procedures. Its primary utility lies in the strategic synthesis of oligosaccharides and glycoconjugates. This compound enables chemists to manipulate galactose frameworks with greater precision, facilitating the introduction of additional functional groups or the coupling to other sugar moieties through controlled glycosylation reactions. The derivative is particularly valuable in the synthesis of targeted glycan structures that are critical for studying carbohydrate functions in biological systems, such as cell signaling and molecular recognition processes. This chemical′s contribution to research extends beyond simple sugar synthesis; it aids in the elucidation of complex biological pathways involving carbohydrates, providing insights into carbohydrate-based interactions. Its application is pivotal in advancing the fundamental understanding of glycoscience, serving as a key reagent in the toolkit of chemists developing new materials and biomolecular architectures.
1,2:3,4-Di-O-isopropylidene-α-D-galacto-hexadialdo-1,5-pyranose (CAS 4933-77-1) References
- Novel oximes as blood-brain barrier penetrating cholinesterase reactivators. | Garcia, GE., et al. 2010. Chem Biol Interact. 187: 199-206. PMID: 20227398
- Synthesis of Glycomimetics by Diastereoselective Passerini Reaction. | Vlahoviček-Kahlina, K., et al. 2018. J Org Chem. 83: 13146-13156. PMID: 30354108
- Glycosylated porphyrin derivatives and their photodynamic activity in cancer cells[J]. | Vedachalam S, Choi B H, Pasunooti K K,. 2011. MedChemComm,. 2(5): 371-377.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
1,2:3,4-Di-O-isopropylidene-α-D-galacto-hexadialdo-1,5-pyranose, 250 mg | sc-222901 | 250 mg | $200.00 |
